pharmaceuticals
A team of biohackers is on a David-versus-Goliath mission to make insulin affordable to an increasing number of diabetics.
Plan S is starting to take hold, but the cost is merely shifting even more to the researchers.
There has been a dramatic increase in abuse and misuse.
We owe a lot to vaccines and the scientists that develop them. But we’ve only just touched the surface of what vaccines can do.
▸
17 min
—
with
The U.S., China, and Russia are in a “vaccine race” that treats a global challenge like a winner-take-all game.
Addiction is not a moral failure. It is a learning disorder, and viewing it otherwise stops communities and policy makers from the ultimate goal: harm reduction.
▸
19 min
—
with
That question is at the heart of the new documentary, “Medicating Normal.”
An article in Journal of Bioethical Inquiry raises questions about the goal of these advocacy groups.
Should pharmaceutical companies pay people for their plasma? Here’s why paid plasma is a hot ethical issue.
▸
17 min
—
with
In 2018, cancer drugs earned the pharmaceutical industry $123.8 billion. Soon, they’ll be worth billions more.
Dr. Charles Grob was the first researcher granted FDA approval to study these drugs.
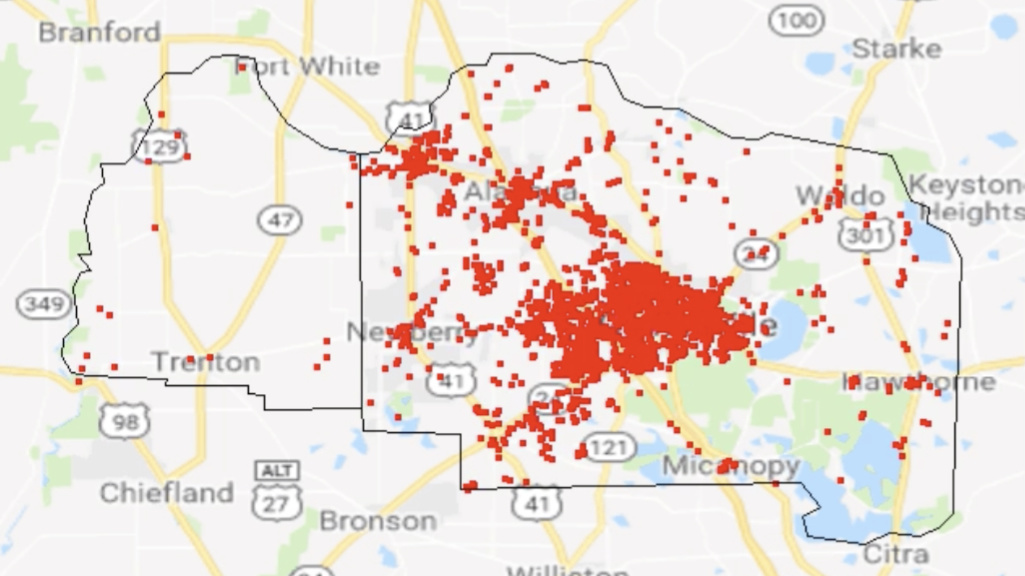
A new study analyzed more than 1.5 billion opioid prescriptions over eight years.
The move reflects a broader nationwide effort to lower prices of the life-saving drug.
Dr. Joel Smithers was recently sentenced to decades in prison for the numerous illegal prescriptions he gave out.
FieldTrip is advancing the realm of clinical psychedelic testing.
With the ivory trade on the decline, poachers have been capitalizing on a new, disturbing trend.
Here’s how we stop a health crisis before it wreaks havoc on us.
▸
15 min
—
with
Even more intriguing is the reason: recognizing facial expressions.
Normally, life expectancy increases every year. For Americans, the opioid epidemic is changing that.
Don’t believe a small reduction in immunization matters?
Anxiety does not help other medical conditions, adding trouble on top of trouble.
Antibiotics are often the fastest and simplest way for doctors to help their patients. But with the threat of drug-resistant bacteria, how do we stop prescribing antibiotics?
It marks a major shift in the government’s battle against the opioid crisis.
When these companies compete, in the current system, the people lose.
▸
5 min
—
with
Alternative treatments are often better for noncancer pain, the study found.
Drug treatment centers pose potential threats to drug addicts.
▸
5 min
—
with
A tech-minded approach to drug fraud could squash those who enable the deadly opioid crisis.
▸
5 min
—
with
The Great White North has found a way to provide universal healthcare with more salubrious results and trimmed national costs. Take notes, America.
The results of a recent trial found that a new oral immunotherapy regimen could have life-saving potential.
Here are the leading solutions to antibiotic resistance, the next major global health threat.
▸
6 min
—
with