marine biology
To date, only one research vessel has ever encountered a milky sea.
A simple trick allowed marine biologists to prove a long-held suspicion.
“Don’t tread on me” is a slogan of the deep sea, too.
The bizarre discovery could pave the way for advances in regenerative medicine for humans.
The famous cognition test was reworked for cuttlefish. They did better than expected.
The organisms were anchored to a boulder 900 meters beneath the ice, living a cold, dark existence miles away from the open ocean.
A new paper explores how noise from human activities pollutes the oceans, and what we can do to fix it.
How do these little beasties detect light anyway?
Exceptionally high-quality videos allow scientists to formally introduce a remarkable new comb jelly.
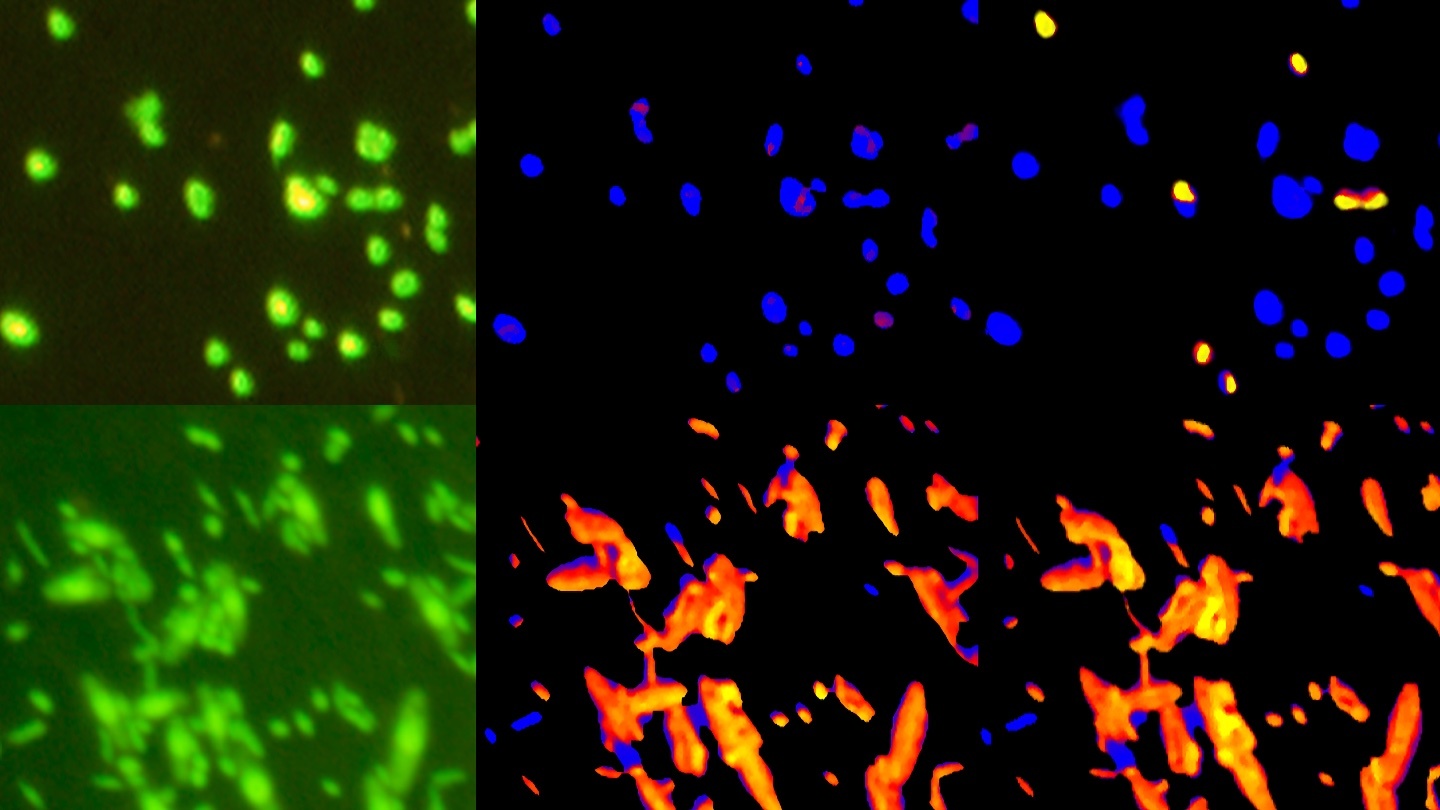
The microbes that eventually produced the planet’s oxygen had to breathe something, after all.
Sharks fear killer whales. How does this impact the ecosystems they share?
A new study finds that starlet sea anemones have the unique ability to grow more tentacles when they’ve got more to eat.
A new study bases its calculations on more than the great white shark.
According to international law, the seabed belongs to everyone.
In one of the ocean’s most lifeless places, scientists discover and resuscitate ancient organisms.
The complacent majority needs to step up and call for action on climate change.
▸
22 min
—
with
These alien-like creatures are virtually invisible in the deep sea.
A new study lays out the case for the damaging effects of stress on orcas living in tanks.
The ocean’s largest shark relies on vision more than previously believed.
Scientists are befuddled by where the shark gets most of its food.
Non-avian dinosaurs were thought terrestrially bound, but newly unearthed fossils suggest they conquered prehistoric waters, too.
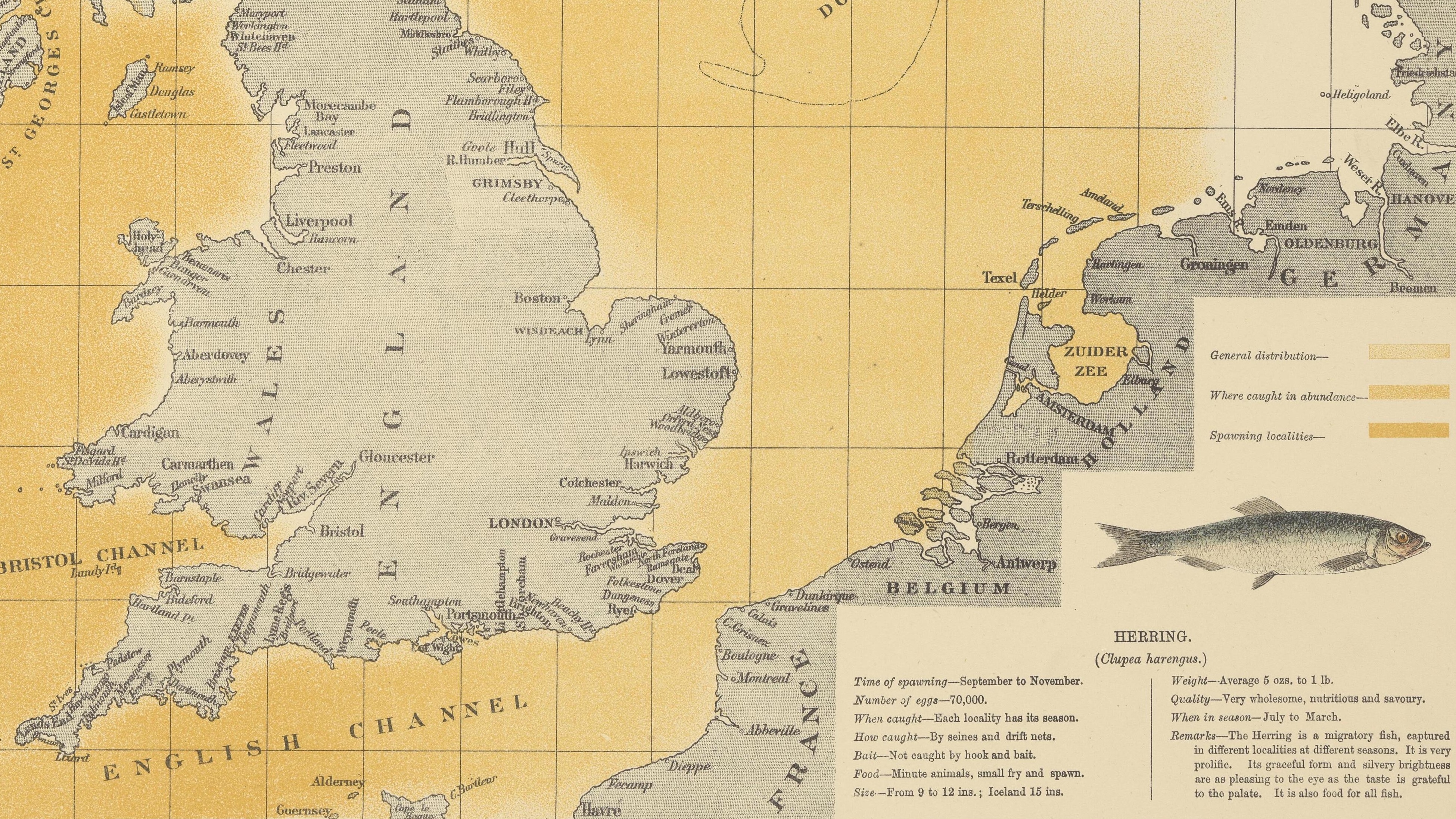
O.T. Olsen’s gorgeous ‘Piscatorial Atlas’ (1883) describes a world now destroyed and forgotten
The relatively quick evolution of nine unusual shark species has scientists intrigued.
The axolotl is known to regrow its lower jaw, its retinae, ovaries, kidneys, heart, rudimentary lungs, spinal cord, and large chunks of its brain.
Let’s not kid ourselves: Coral reefs are in serious danger. But numerous ambitious projects are underway with the goal of keeping these ecosystems alive.
Octopuses are known to rapidly change colors during sleep, but it’s still unclear whether they dream like humans do.
Humpbacks swap songs at remote group of islands in the South Pacific.
It’s only the second pocket shark specimen ever discovered.
New research reveals a major shift in what pressures life used to face.
A comprehensive interdisciplinary paper removes any doubt that orcas don’t belong in marine parks and zoos.