invention
Virtual reality continues to blur the line between the physical and the digital, and it will change our lives forever.
Thomas Baldwin’s Airopaidia (1786) includes the earliest sketches of the earth from a balloon.
How fabric helped build modern civilization.
▸
6 min
—
with
The EmDrive turns out to be the “um…” drive after all, as a new study dubs any previous encouraging EmDrive results “false positives.”
Inventions with revolutionary potential made by a mysterious aerospace engineer for the U.S. Navy come to light.
Meet a spectacular new blue—the first inorganic new blue in some time.
Researchers find a way to distort laser light to survive a trip through disordered obstacles.
The satellite would burn instead of becoming more space debris.
Researchers from MIT invent a highly accurate clock using quantum entanglement that can lead to new physics.
An important step toward figuring out our space station future.
Scientists at Washington University are patenting a new electrolyzer designed for frigid Martian water.
A clever new design introduces a way to image the vast ocean floor.
Australian researchers figure out a new way to apply extreme pressure and squeeze out diamonds.
Can passenger airships make a triumphantly ‘green’ comeback?
Researchers design microdevices that can gradually deliver medicine by latching on to intestines.
While it’s always been a boon to Popeye’s “muskles,” it looks like spinach may also have a role to play in clean future batteries.
New experiments find weird quantum activity in supercold gas.
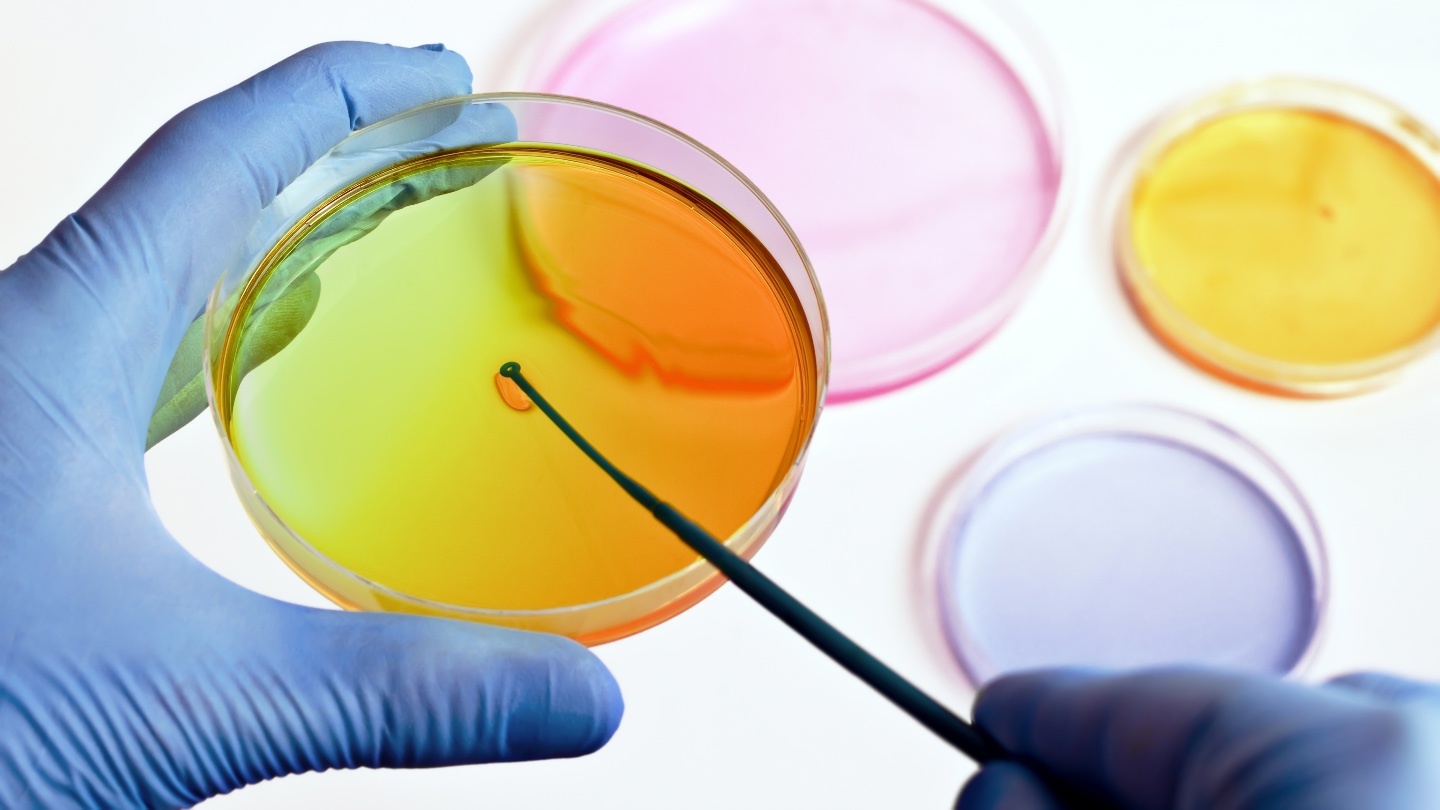
New prototype Petri dishes let ordinary scientists in on the advanced technology.
Dust sticking to things on the moon is a serious problem researchers are trying to solve.
Utilizing nuclear waste converted to diamonds, this company’s batteries will reportedly last thousands of years in some cases.
A clever new study definitively measures how long it takes for quantum particles to pass through a barrier.
The space tourism company Virgin Galactic teams up with Rolls Royce to create a new Mach 3 supersonic aircraft.
Smart bandages quickly identify antibiotic-resistant bacteria, and normal bacteria, in owies.
By leveraging the difference between lit and shadowed areas, a new energy source perfect for wearables is invented.
Astrophysicist Michelle Thaller talks ISS and why NICER is so important.
▸
5 min
—
with
Researchers devise groundbreaking new methods to create and duplicate single-atom transistors for quantum computers.
Why did the dinosaurs go extinct? Because they didn’t have a space program.
▸
4 min
—
with
He’s also credited by some as having coined the phrase “user-friendly.”
The smart toilet can analyze urine and stool samples for disease markers and can even recognize an individual user’s “analprint”.
Using permanent magnets may help to make nuclear fusion reactors simpler and more affordable.