medical research
David Julius and Ardem Patapoutian were awarded the highest honor in medicine for their research into how human bodies make sense of and respond to the outside world.
A new study looks at what happens when you get infected with two viruses at the same time.
MIT researchers design glue that mimics the sticky substance barnacles use to cling to rocks.
The design could help restore motor function after stroke, enhance virtual gaming experiences.
The risk of dying basically flattens after age 110.
A new study found that people who scored high in certain psychopathic traits are more likely to limit head movements.
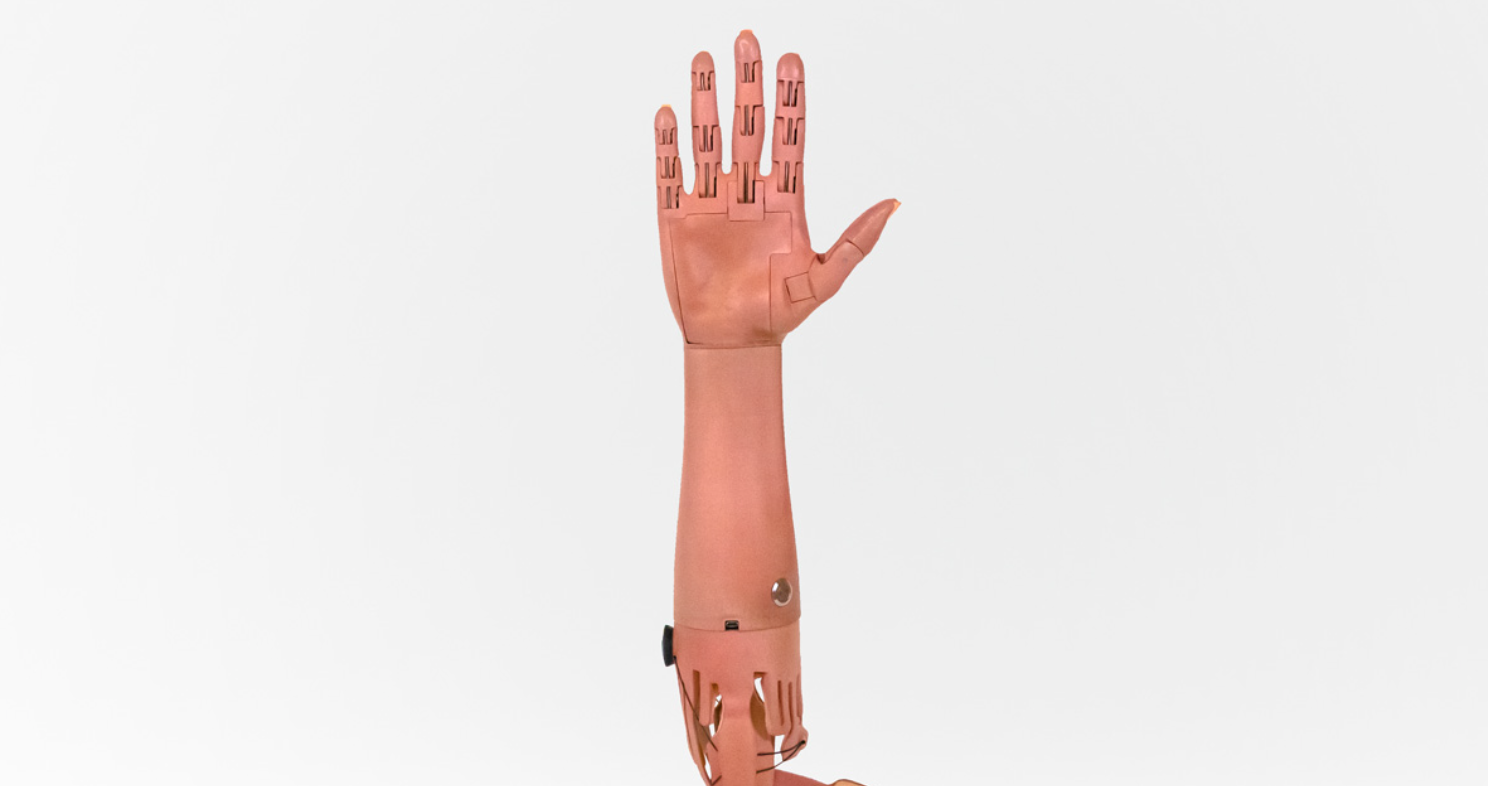
Prosthetic arms can cost amputees $80,000. A startup called Unlimited Tomorrow is aiming to change that by making customized 3D-printed bionic arms for just $8,000.
Cancer cells seem to have a harder time growing among pair-bonded mice, according to a new study that explored the “widowhood effect.”
Age ain’t nothing but a number, but “inflammatory age” may be real.
New research shines a light on the genetics of sudden cardiac deaths.
A new device cured the hiccups 92 percent of the time in a recent study involving more than 200 participants.
It could lead to a massive uptake in those previously hesitant.
Laughing gas may be far more effective for some than antidepressants.
An early feasibility study finds a potential new treatment for Alzheimer’s disease.
The treatment is here, but are we ready?
Can the main psychoactive ingredient of magic mushrooms help treat the world’s sixth most debilitating illness?
Who needs steroids when you have the placebo effect?
A new study used functional near-infrared spectroscopy (fNIRS) to measure brain activity as inexperienced and experienced soccer players took penalty kicks.
Want to live 100+ years? You may need unusually good DNA repair.
Healthy people need healthy microbiomes from an early age.
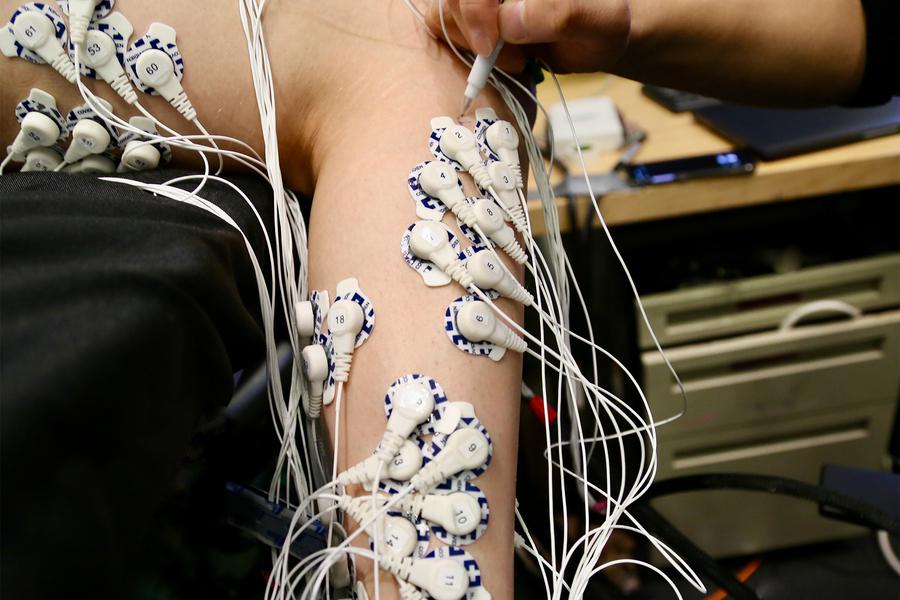
How can researchers map something as complex as the human brain?
Are “humanized” pigs the future of medical research?
The conventional wisdom may be wrong. Consulting Google for information about medical symptoms might not be as counterproductive as commonly thought, new research suggests.
Awareness of one’s own heartbeat has some positive effects.
The retraction crisis has morphed into a citation crisis.
The new treatment targets the underlying genetic cause of the disease.
Reconnecting muscle pairs allows for better sensory feedback from the limb.
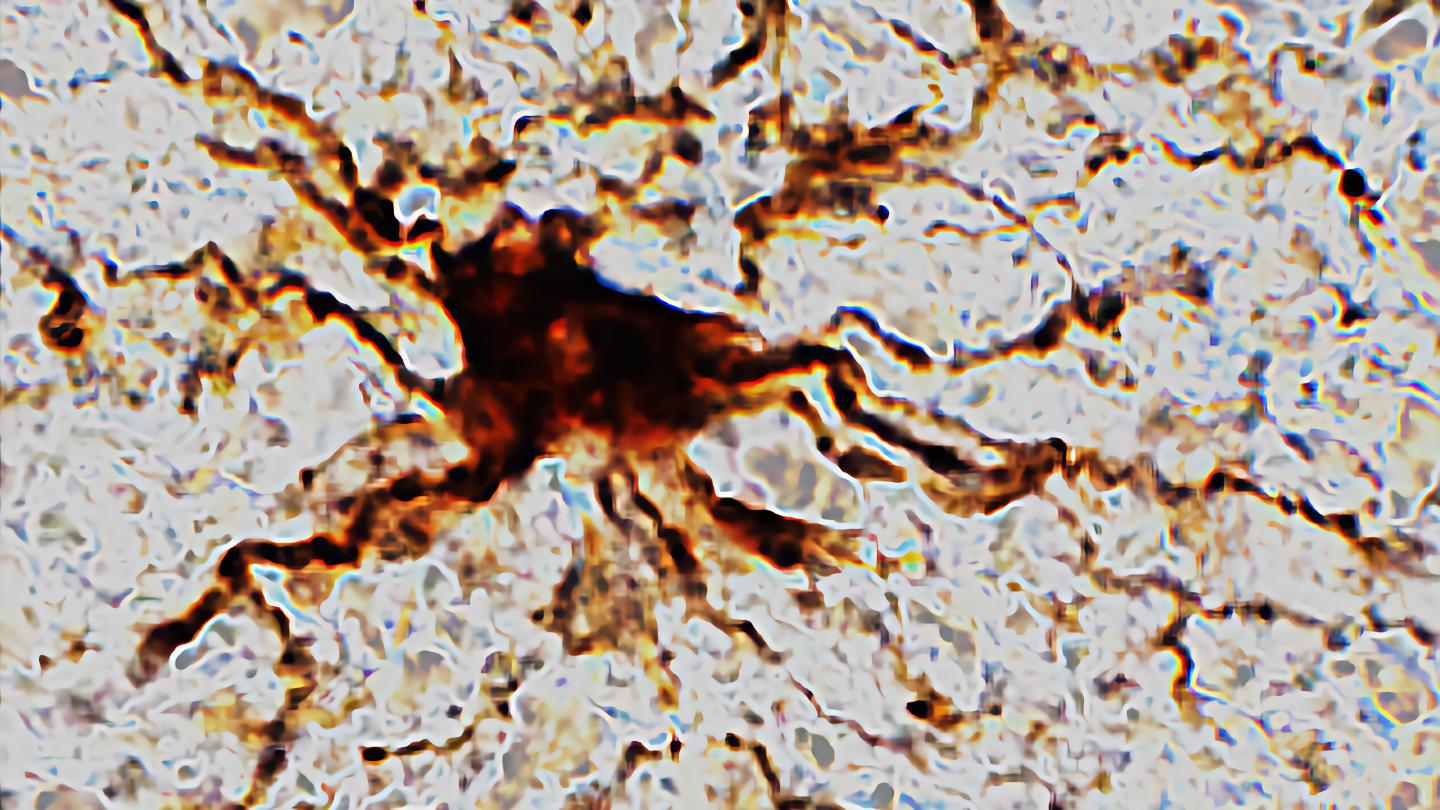
Researchers at the University of Illinois Chicago find that death triggers increased activity in certain brain cells.
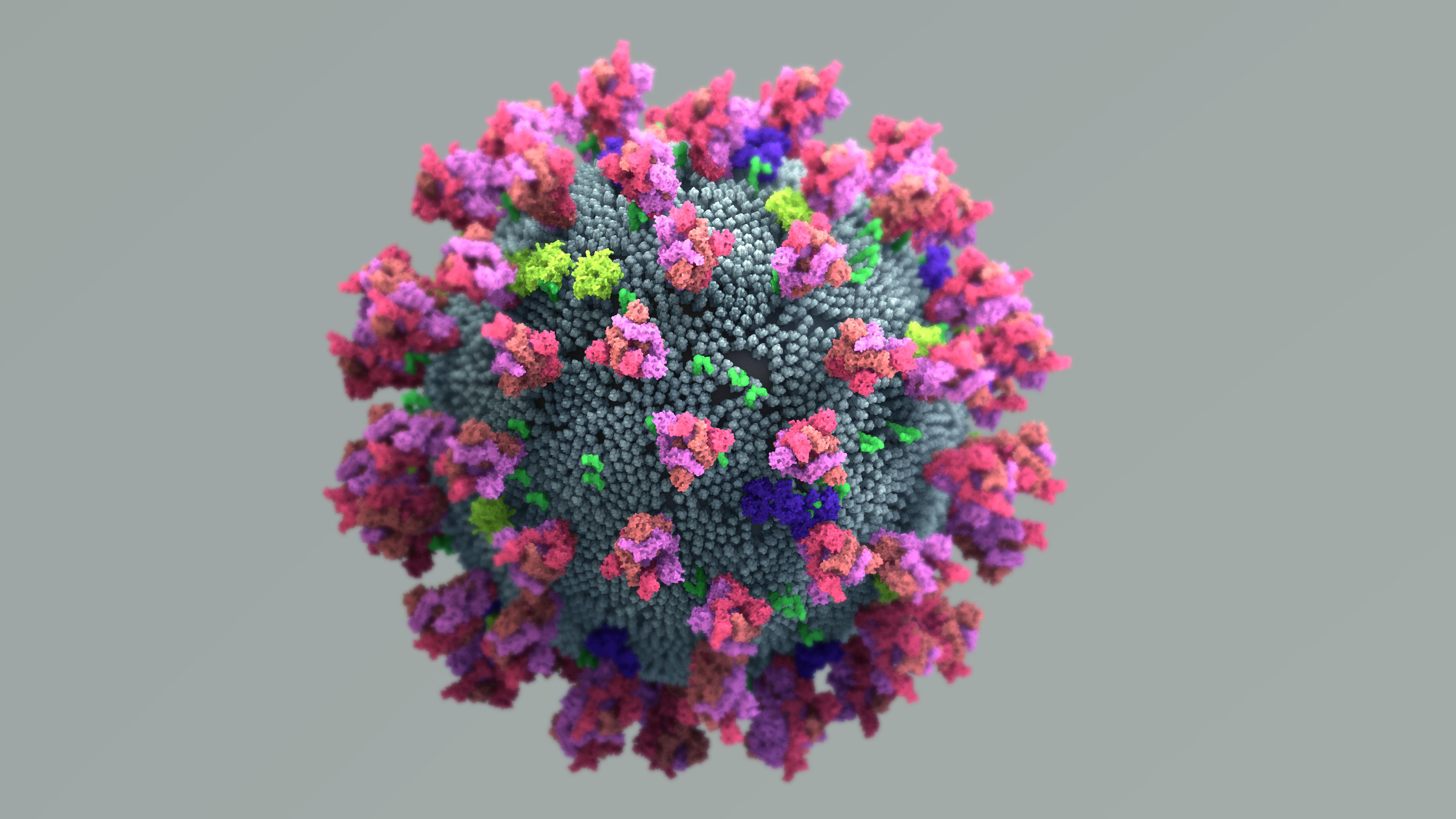
Ultrasound might be able to damage the novel coronavirus in the same way an opera singer’s voice can shatter a wine glass.
Cotton mask fibers prove 33 percent more effective at blocking viruses in trials.