reproduction
Evolutionary success is not about the number of one’s children, but one’s grandchildren: the children need to survive and pass on their genes.
The tiny swimmers appear to transmit signals that “persuade” the female body to have a baby.
Imagine poisoning your rival and yourself and giving only yourself the antidote.
Previous research suggesting it’s all about prolactin may be missing the mark.
Monogamy is often considered a key component of traditional marriages, but it’s only half the story.
▸
8 min
—
with
Positive, romantic thoughts could produce positive, romantic outcomes while dating.
New research reveals that because of an optical illusion, we’ve been viewing sperm incorrectly for nearly 350 years.
You may be surprised at how your body and brain react to this type of pleasure.
Life forms on Earth are wildly varied, but scientists discover a single formula that predicts every one’s life cycle.
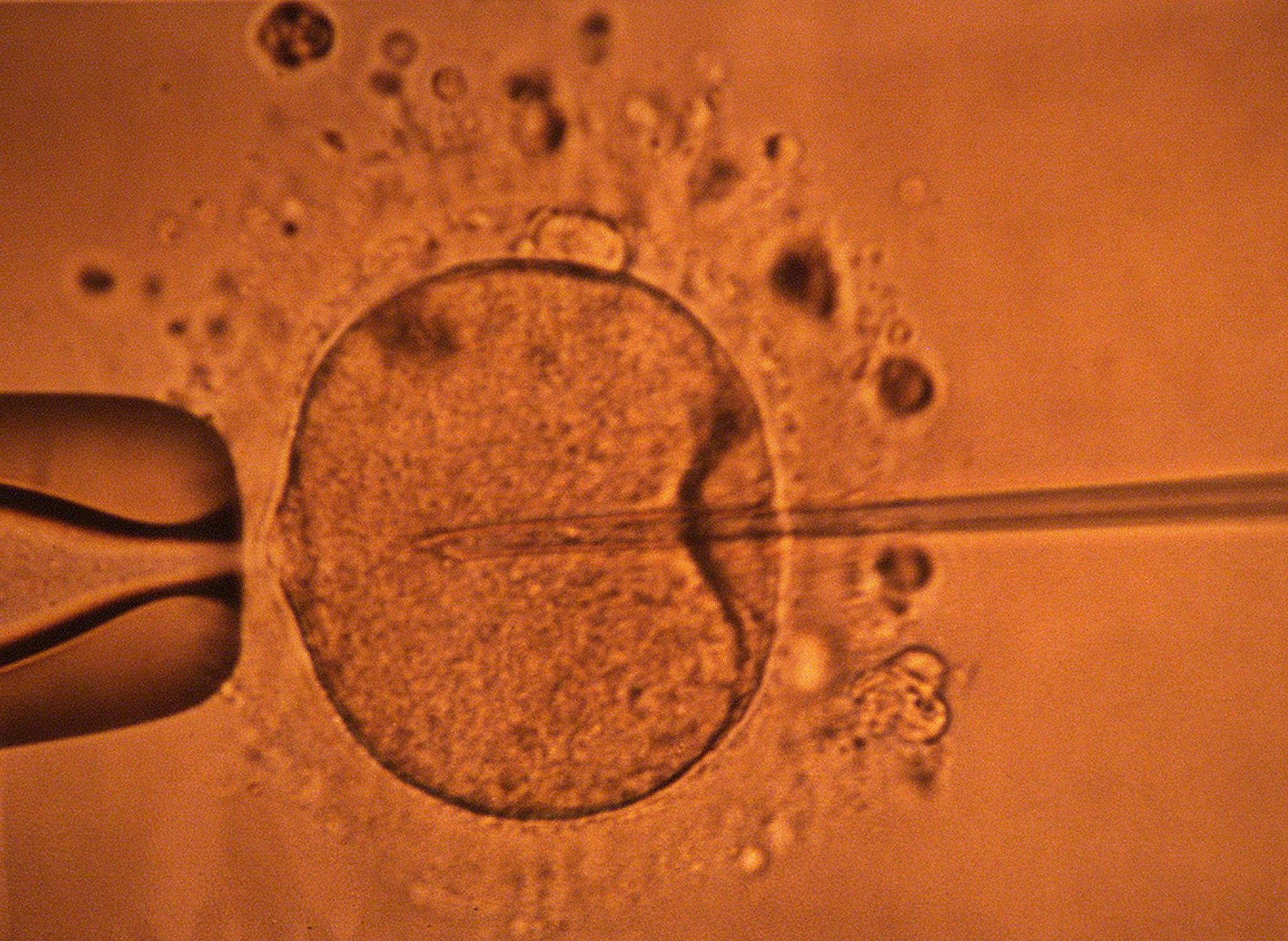
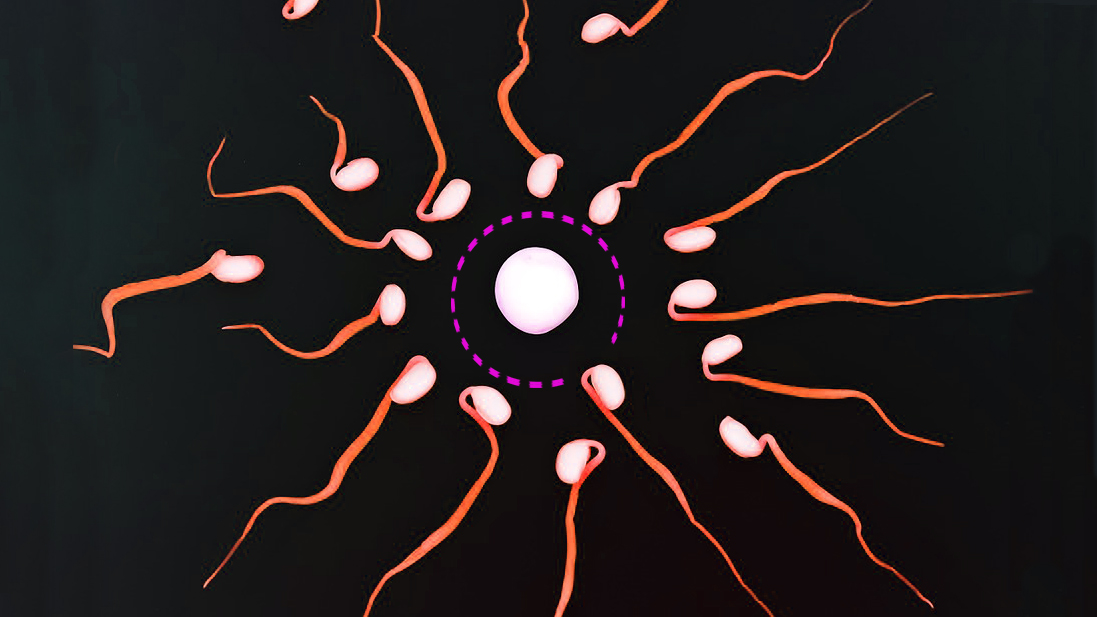
Here’s the first evidence to challenge the “fastest sperm” narrative.
AI is capable of self-reproduction—should humans be worried?
▸
6 min
—
with