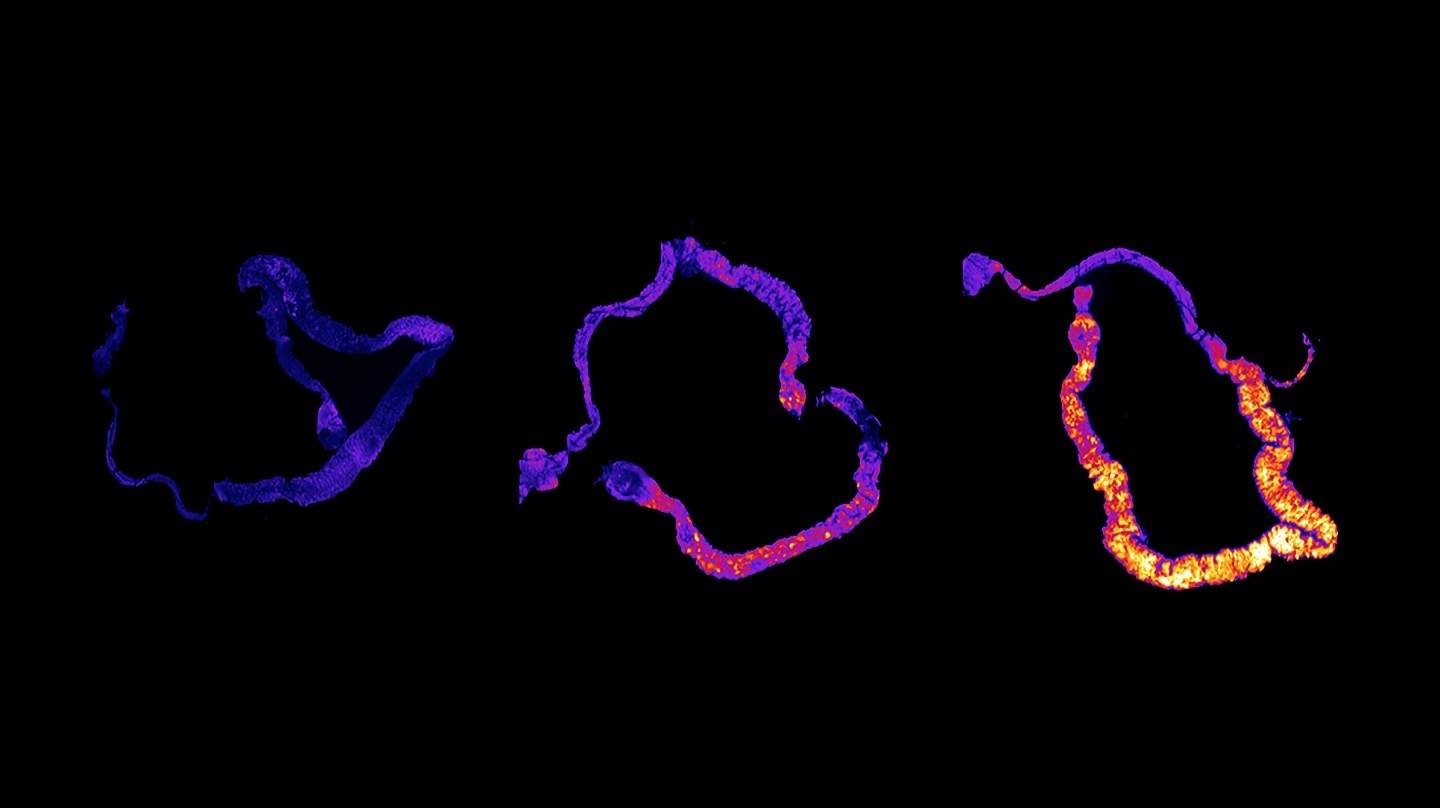molecular biology
New research establishes an unexpected connection.
Dr. Kate Biberdorf explains why boiling water makes it safer and how water molecules are unusual and cool.
▸
3 min
—
with
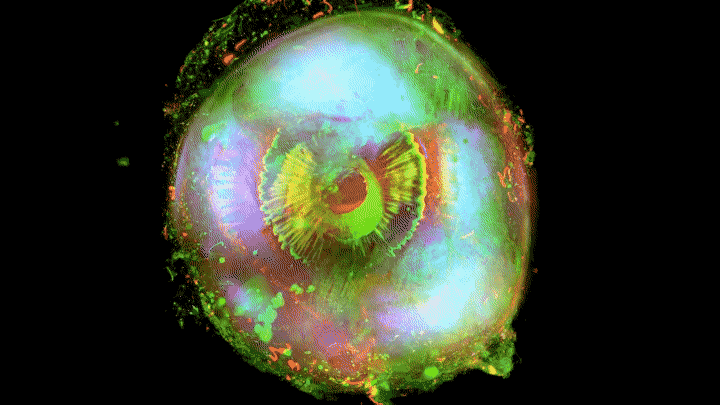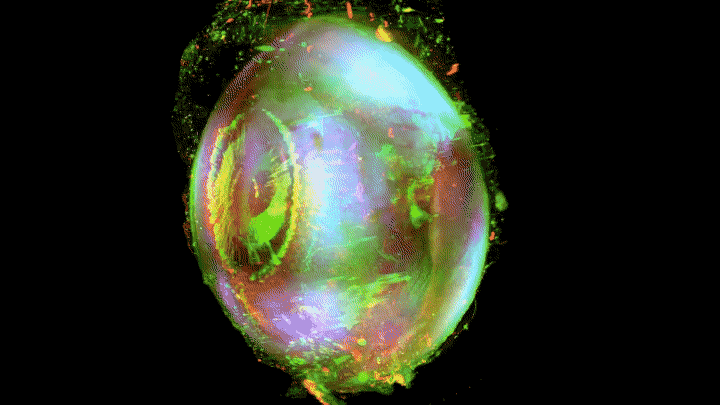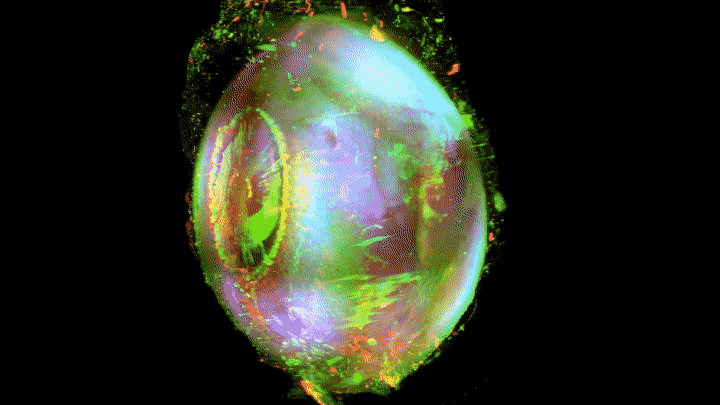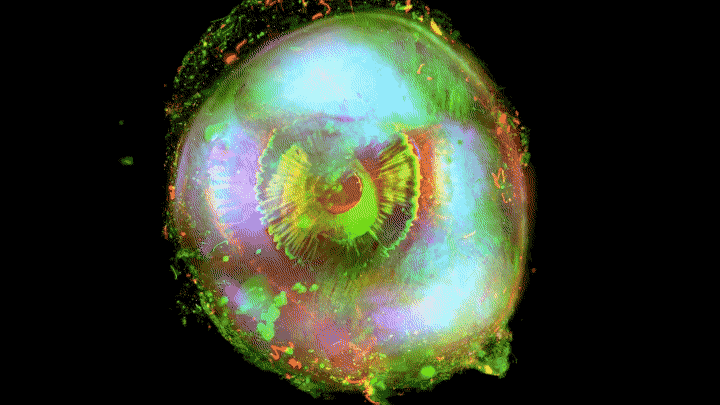
Add some color to the internal structures and you’ve got some eye-popping imagery.
Scientists figured out how a certain treatment for skin cancer gives some patients a visual “superpower.”
A recent computer analysis found that millions of possible chemical compounds could be used to store genetic information. This begs the question — why DNA?
Scientists speculate that if life were to have spontaneously developed on Earth, the first thing there would need to be are vesicles.
NASA’s intrepid Curiosity rolls upward into Mars history.
Three scientist friends, working separately, share the prestigious prize.
Cancer’s sweet tooth. Turning cancer cells into fat. Unveiling genetic secrets. Scientists are learning about cancer every day.
Here’s why you might eat greenhouse gases in the future.
Stems cells have always been pretty amazing.
Doctors may be able to painlessly reshape cartilage with the technique.
Artistic depictions of the atom have deceived us all.
▸
3 min
—
with
Researchers at the University of Basel in Switzerland have hijacked cancer’s cellular plasticity to turn the disease against itself.
Patients with lung disease could find relief by breathing in messenger RNA molecules.
MIT researchers have discovered how to turn wasp venom into an antibiotic.
Astronauts will be able to harvest the Moon’s natural resources to sustain human life.
▸
3 min
—
with
As costs go down and the benefits become more clear, can we afford not to eat lab-grown meat?
NASA research finds a new direction in searching for signs of life in the Universe.