health care
Longevity gets a new motto: location, location, location.
Laughing gas may be far more effective for some than antidepressants.
Healthy people need healthy microbiomes from an early age.
Are “humanized” pigs the future of medical research?
The neoliberal call for more ‘choice’, seems hard to resist.
As patients approached death, many had dreams and visions of deceased loved ones.
Light-emitting tattoos could indicate dehydration in athletes or health conditions in hospital patients.
Beyond making up 70% of the world’s health workers, women researchers have been at the cutting edge of coronavirus research.
Trained dogs can detect cancer and other diseases by smell. Could a device do the same?
The majority of Americans are stressed, sleep-deprived and overweight and suffer from largely preventable lifestyle diseases such as heart disease, cancer, stroke and diabetes. Being overweight or obese contributes to […]
This digital otoscope features a tiny camera and LED lights to help you remove stubborn wax more efficiently.
Most people believe you can win an argument with facts – but when “facts” are so often subject to doubt, are personal experiences trusted more?
Could we have predicted COVID-19 through social media trends?
Scientists regenerate damaged spinal cord nerve fibers with designer protein, helping paralyzed mice walk again.
The long-term lessons America learns from the coronavirus pandemic will spell life or death.
▸
3 min
—
with
Northwell Health is using insights from website traffic to forecast COVID-19 hospitalizations two weeks in the future.
Porcine gene edits may allow such transplants without rejection.
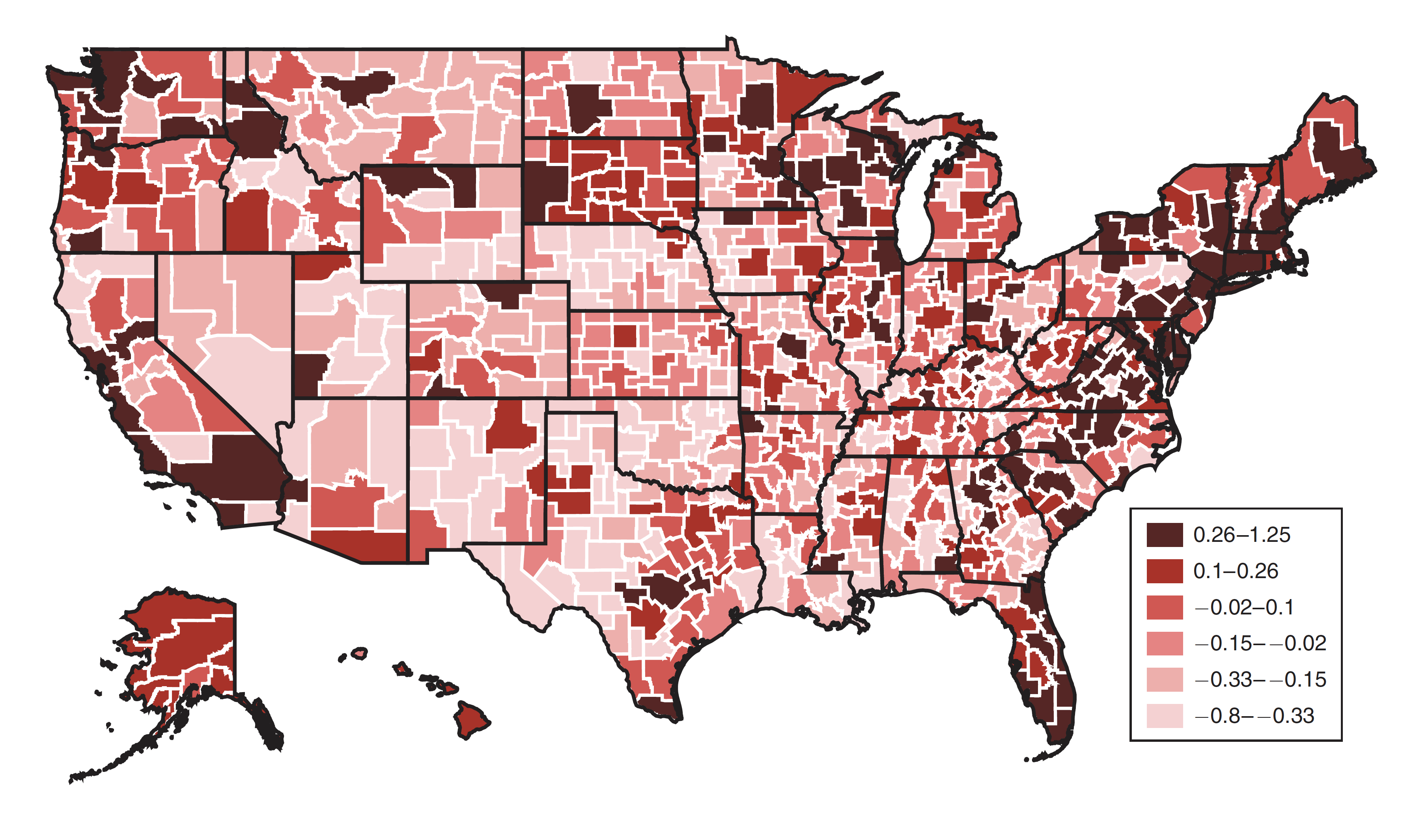
Want to tell someone’s future in the US? You don’t need a crystal ball, just their zip code.
The federal government and private insurers greatly increased Americans’ telehealth access during the pandemic. Will these changes be permanent?
Gun violence is a public health crisis that is notoriously difficult to study because of politics. Finally, a new research initiative has the green light to collect life-saving data.
New research spotlights how low-income Black households face greater financial distress and vulnerability as a result of the pandemic economic crisis.
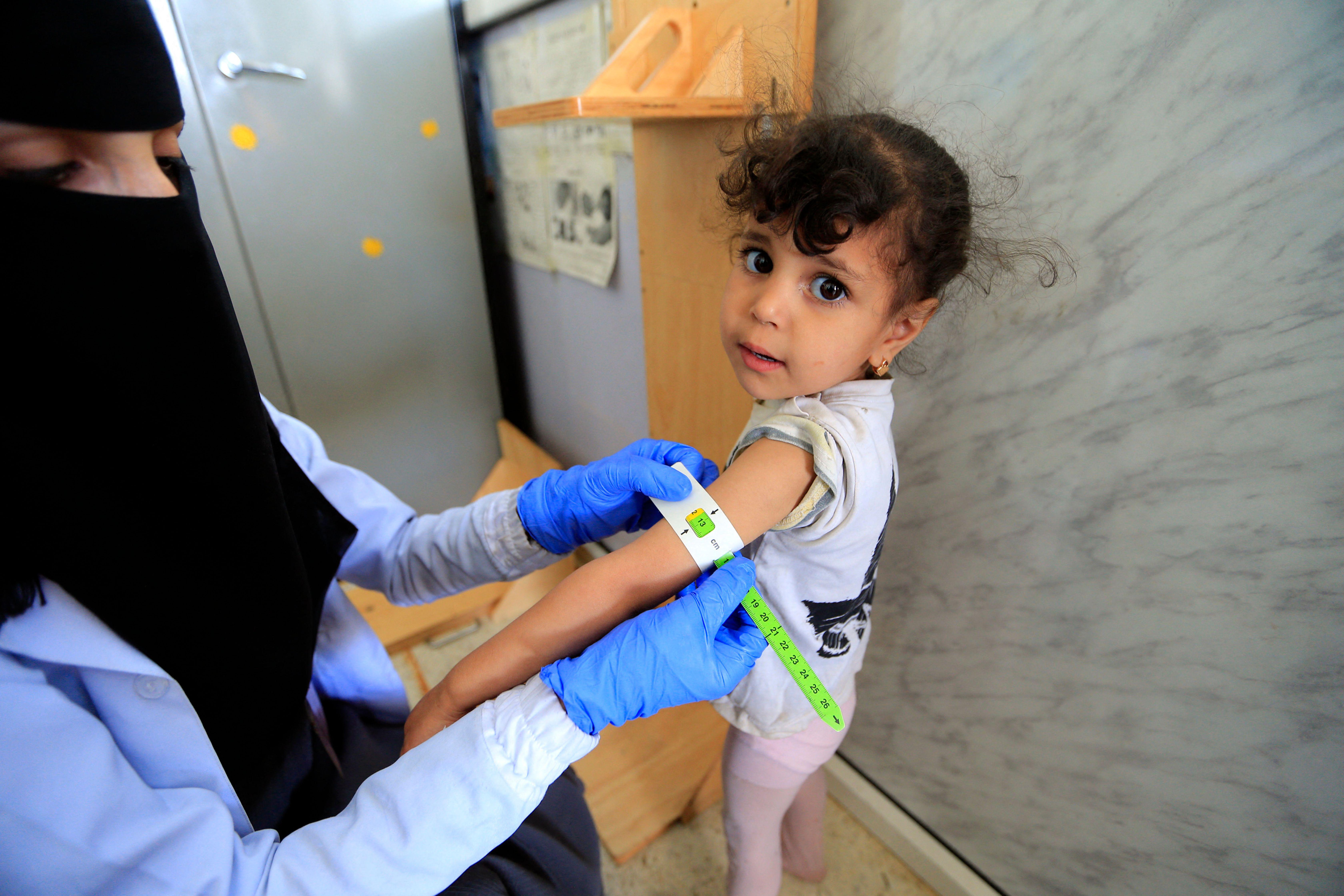
The COVID-19 pandemic is making health disparities in the United States crystal clear. It is a clarion call for health care systems to double their efforts in vulnerable communities.
Your health and the health of the planet are not indistinguishable.
A new study of nurses shows the importance of sleep—and staying aware on the job.
There has been a dramatic increase in abuse and misuse.
Duke University researchers might have solved a half-century old problem.
An algorithm may allow doctors to assess PTSD candidates for early intervention after traumatic ER visits.
An article in Journal of Bioethical Inquiry raises questions about the goal of these advocacy groups.
Innovative drugs are sometimes held up due to old-fashioned human biases.