synthetic biology
We eat 50 billion chickens every year. Is there a better way?
Metal-like materials have been discovered in a very strange place.
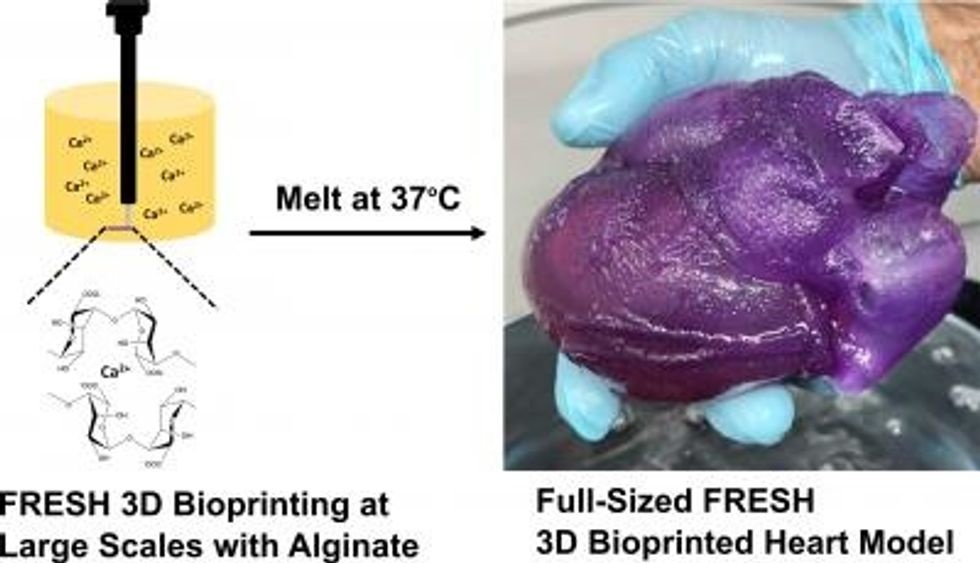
A new method is able to create realistic models of the human heart, which could vastly improve how surgeons train for complex procedures.
DNA molecules are highly programmable.
Exploring how a small change in your DNA sequence can make you a natural blonde.
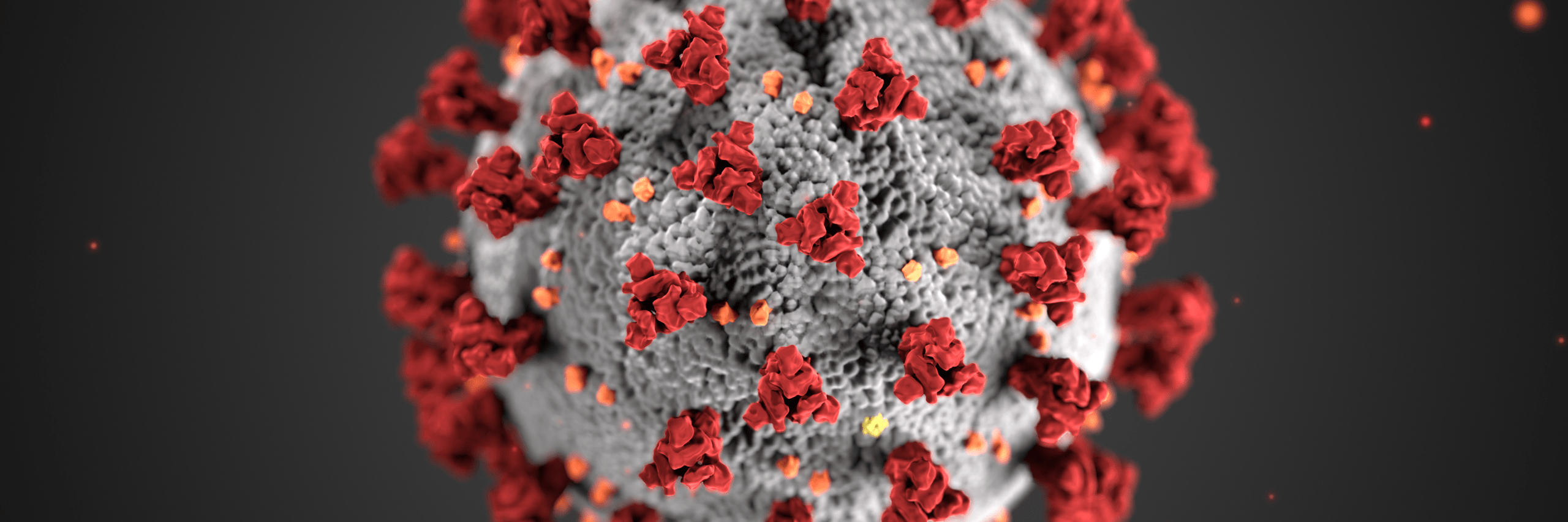
The National Institutes of Health hopes synthetic biology can engineer vaccines that outperform nature.
A recent computer analysis found that millions of possible chemical compounds could be used to store genetic information. This begs the question — why DNA?
The FDA calls out creators of genetically tweaked hornless bulls.
A new report sees a major disruption in where we get our food.
Professor George Church creates a gene “wishlist” that can lead to superhuman abilities.
A unique 3D model allows researchers to explore embryonic development.
Our clever human hands may soon be outdone.
Synthetic biology is changing the way the planet works.
What gives us color now may give rise to our cyborg future.
A new method of growing mini-brains produces some startling results.
Using a new process, a mini-brain develops retinal cells.
As costs go down and the benefits become more clear, can we afford not to eat lab-grown meat?