biotech
Hawking’s refusal to upgrade his communication system preserved a voice that became iconic, not just for its sound, but for the profound identity it conveyed.
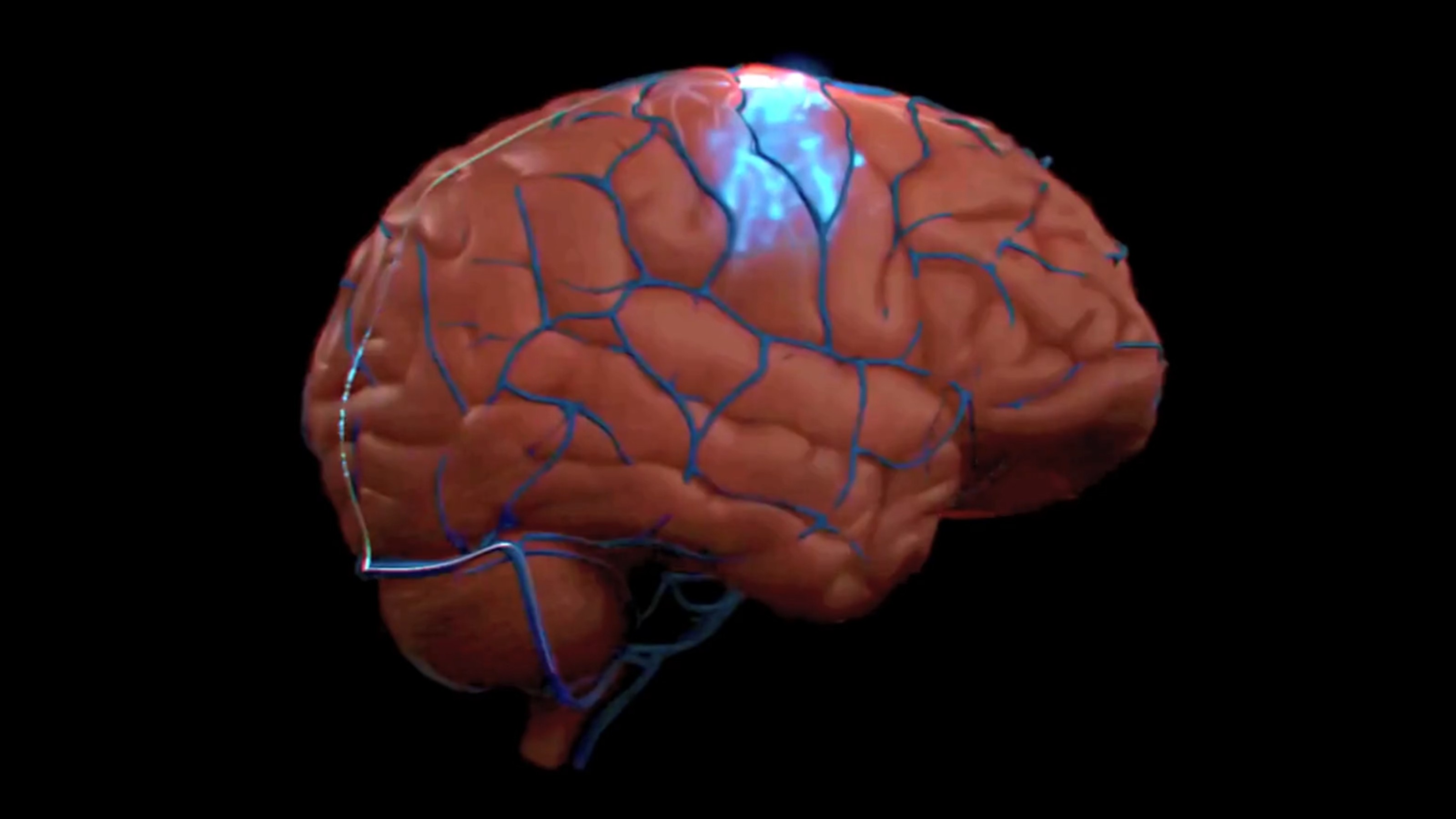
“Neurotech is not just about the brain,” says Synchron CTO Riki Banerjee, explaining how their tech can help with paralysis, brain diseases, and beyond.
Of the millions of substances people encounter daily, health researchers have focused on only a few hundred. Those in the emerging field of exposomics want to change that.
In the international competition, people with physical disabilities put state-of-the-art devices to the test as they race to complete the tasks of everyday life.
“The promise of the Human Genome Project has finally arrived.”
How technology could change everything we thought we knew about reproduction.
There is one obstacle that reliably blocks innovative ideas: how we fund science.
Veteran investor Sujal Patel, co-founder and CEO of Nautilus Biotechnology, helps us sift golden nuggets from the loose shale of entrepreneurship.
Joe Betts-LaCroix — co-founder and CEO of Retro Biosciences — talks to Big Think about invention, authenticity, and Sam Altman’s “art of the startup.”
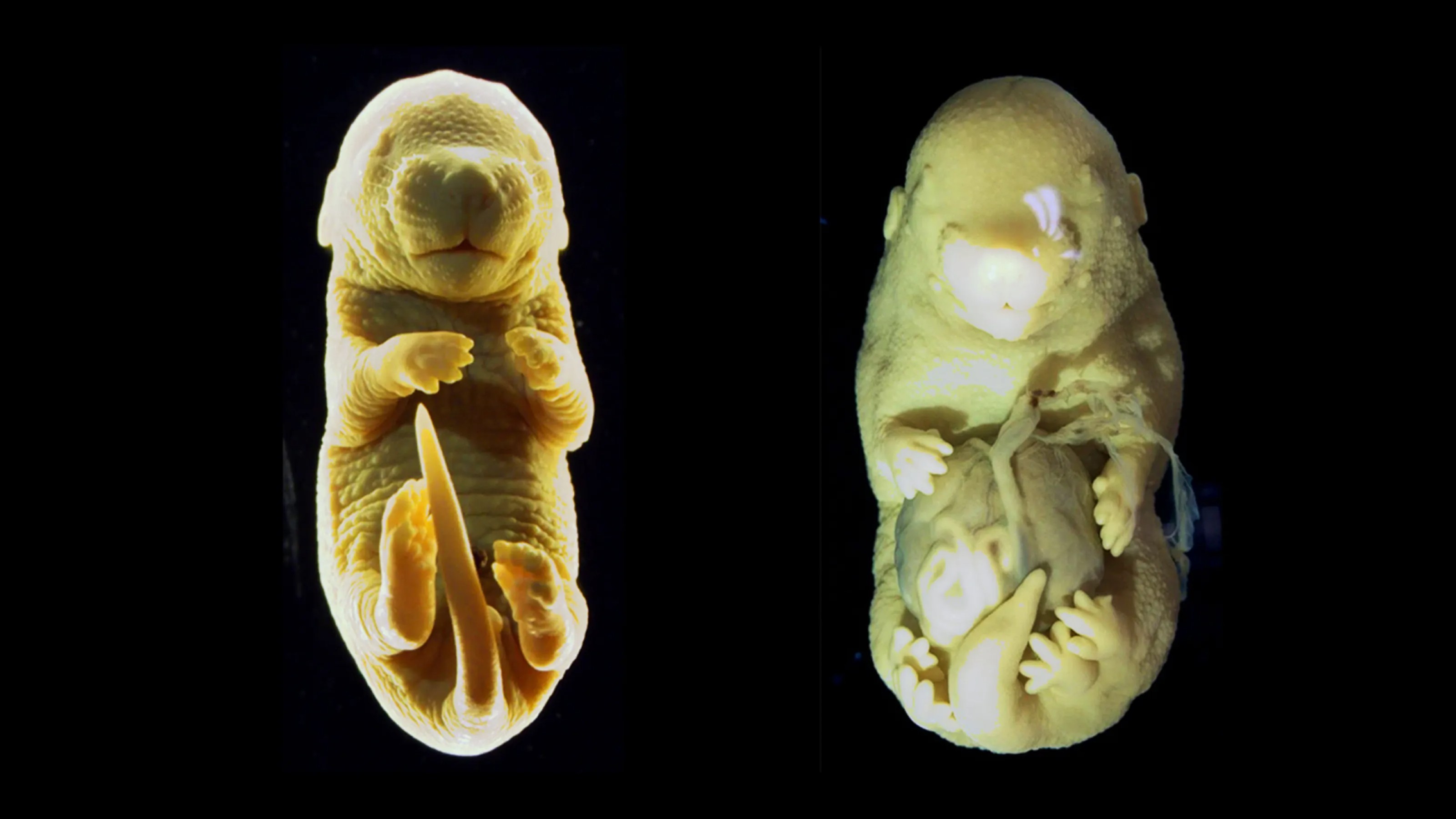
A study of spinal development took a strange turn and made a surprise discovery.
What would it take to create a truly intelligent microbot, one that can operate independently?
The sober reality behind the effectiveness of two new drugs touted as Alzheimer’s breakthroughs: lecanemab and donanemab.
The brain-computer interface will be tested in a six-year trial in patients with quadriplegia.
It’s not just fun: DNA origami has the potential to revolutionize engineering at the nanoscopic scale.
The first human trial of base editing delivered strong results along with some safety concerns.
Artificial intelligence can forecast the behavior of viruses and quickly make vaccines to thwart them.
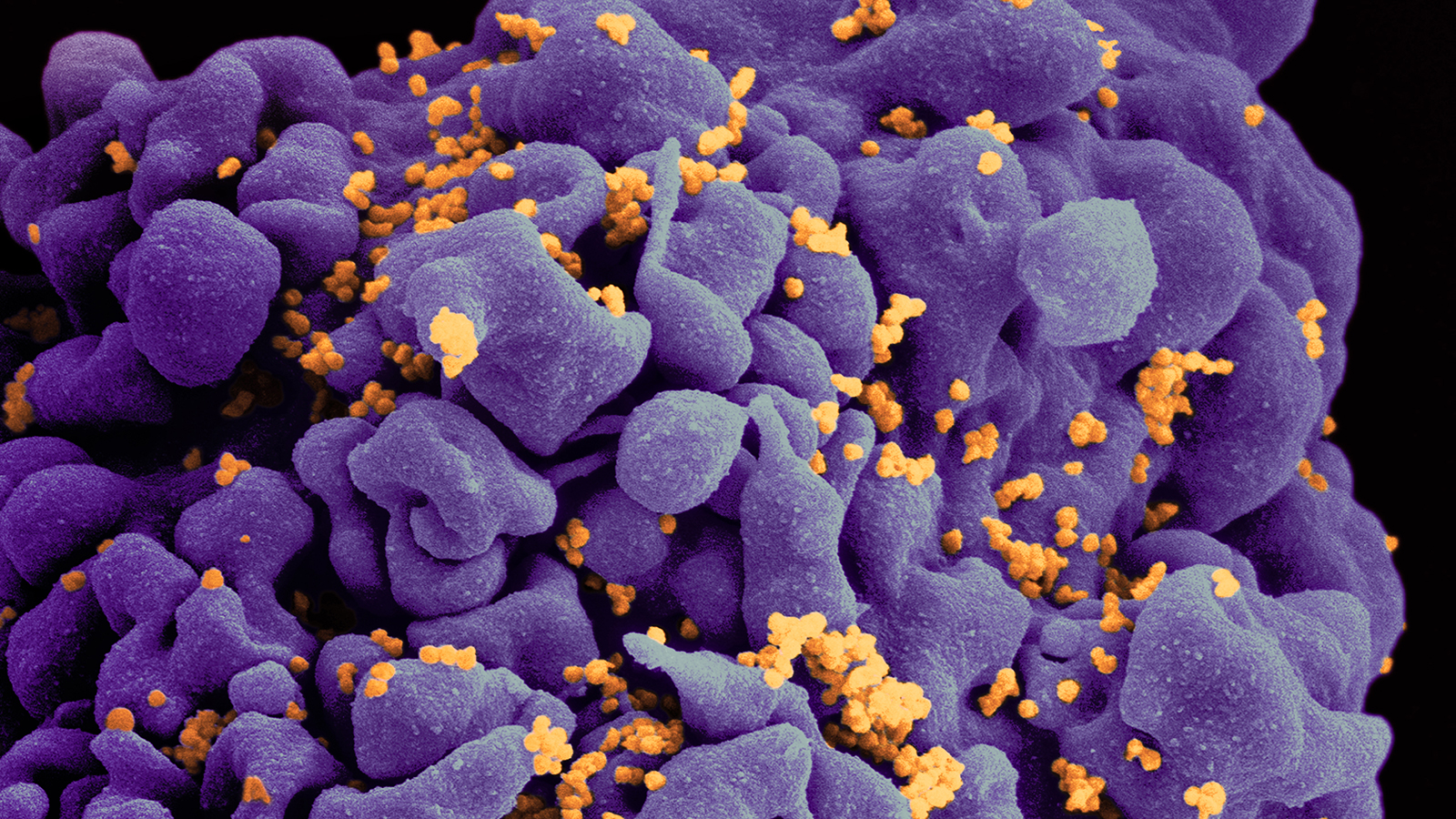
A cure may be on the horizon.
EBT-101 is not the only candidate for an HIV cure. Stem cell transplants, medications, and other CRISPR therapies are being researched.
AI was key to making Moderna’s COVID mRNA vaccine. Its role in mRNA therapeutics will rapidly grow in the coming years.
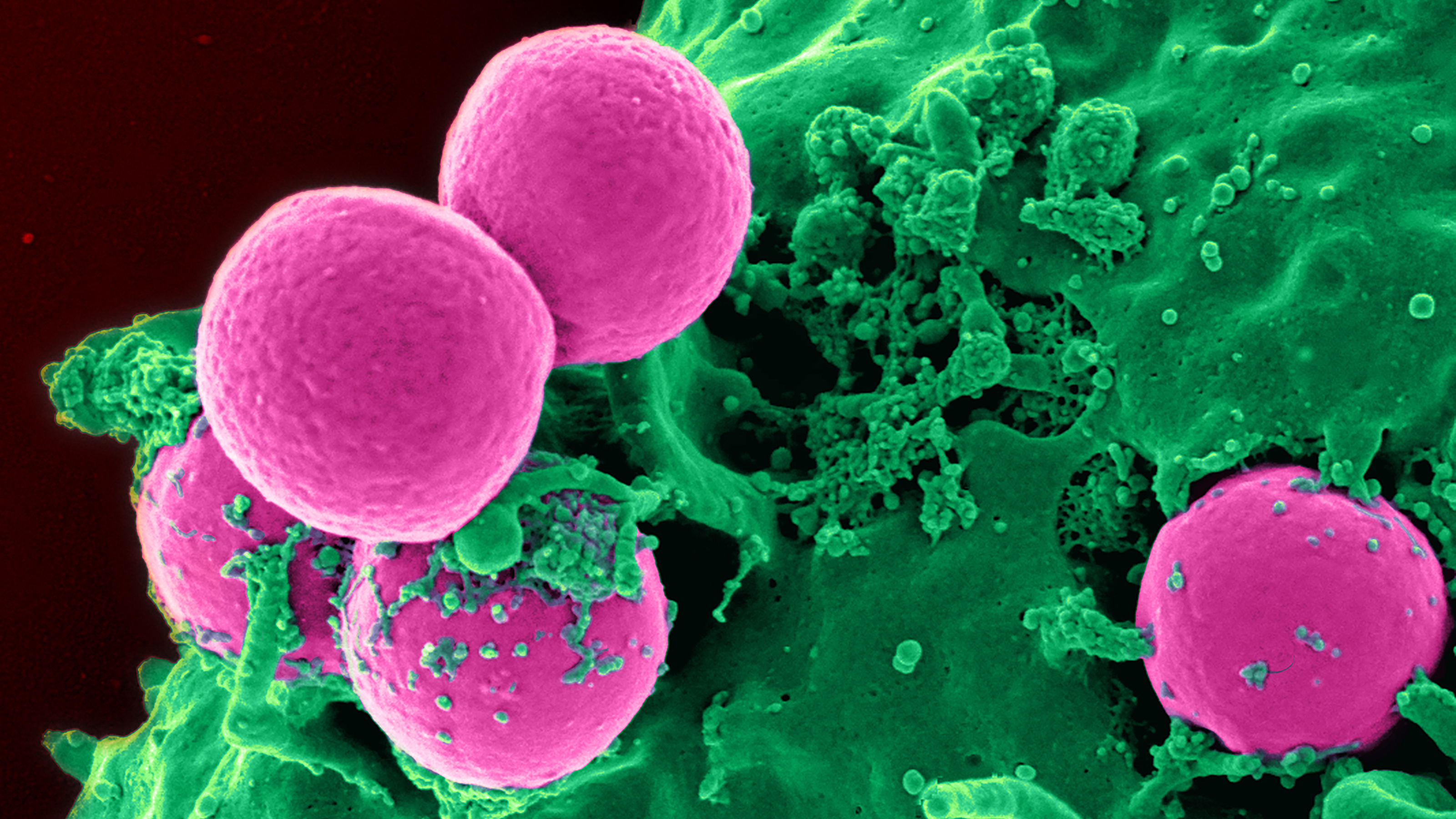
It temporarily puts the immune system on high alert to prevent MRSA, pneumonia, and other infections in the hospital.
You can’t farm spiders — but putting spider genes into silkworms works even better.
Long thought a pipe dream, scientists have discovered a drug that mimics the effects of exercise.
The potential benefits of returning the thylacine to Australia make the project worth the effort.
Undeterred by years of failure, Katalin Karikó and Drew Weissman proved that mRNA is the future of vaccines.
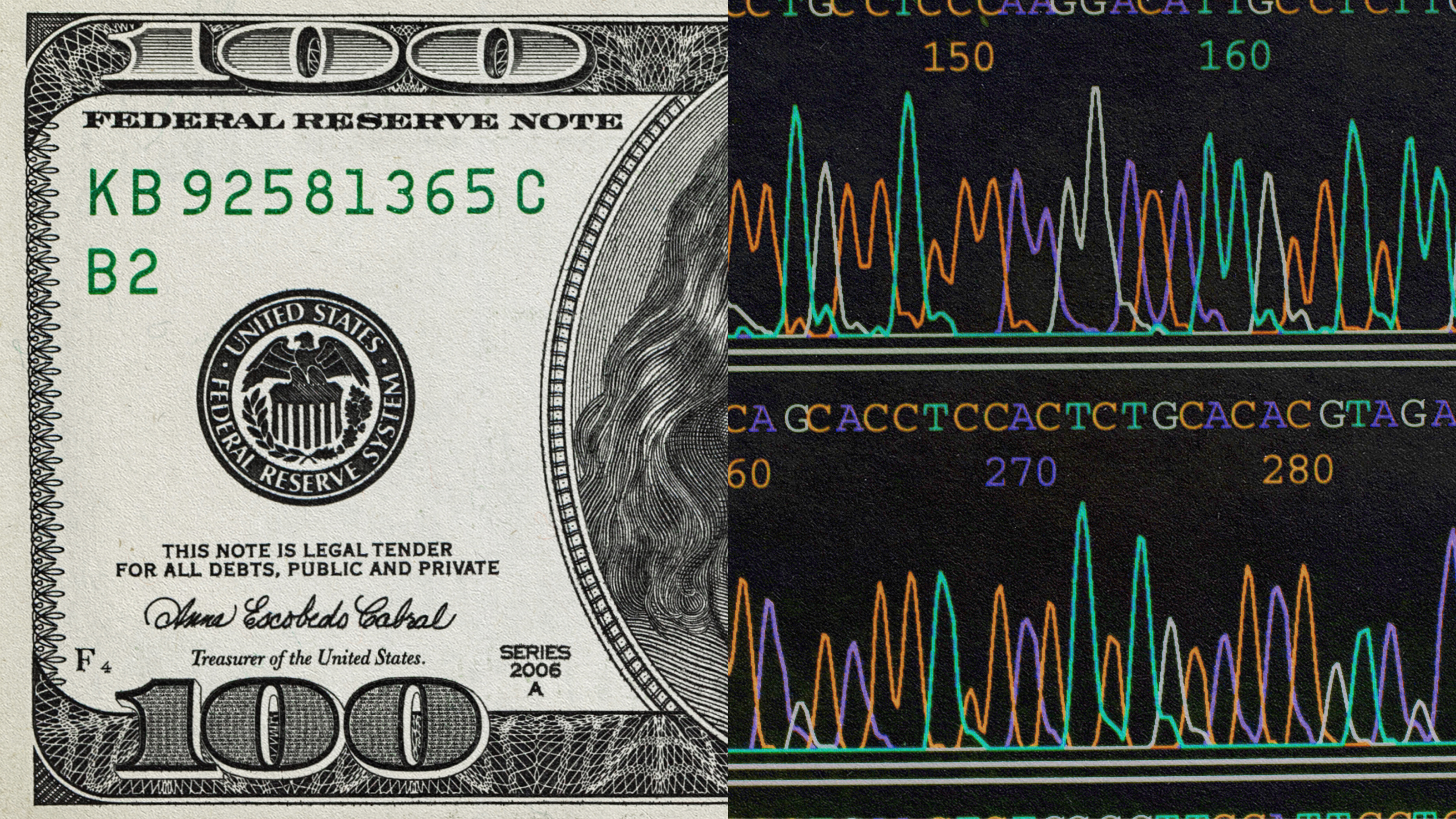
To put things in perspective, the cost of sequencing a single genome in 2012 was around $10,000.
CRISPR, stem cells, and even cancer drugs are helping shape an AIDS-free future.
Subtle clues emerge ahead of the attack via changes in scent.
The brain implant lets her talk four times faster than the previous record.
Today’s popular weight-loss drugs could soon be joined by brain stimulation and gene therapies.
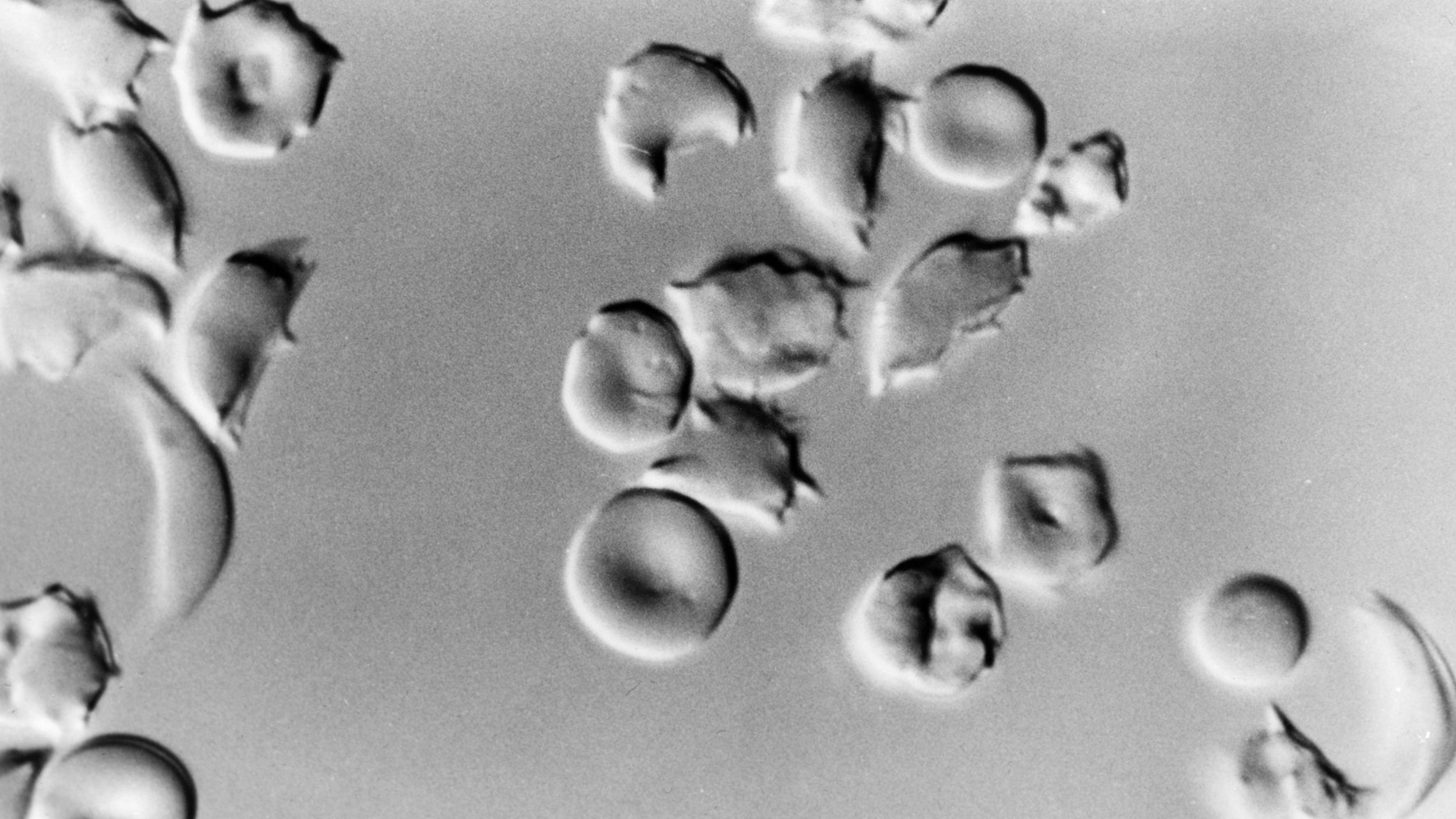
“They decreased their drinking to the point that it was so low we didn’t record a blood-alcohol level.”