chemistry
“I want to change the way we think about the past altogether,” says Dr. Betül Kaçar, an astrobiologist who studies the origin of life.
We’re all entitled to our own opinions, no matter how ill-informed they are. But facts are facts; we can’t just choose the ones we prefer.
One alchemist’s search for a whiz-bang method to produce gold unlocked the central science instead.
It’s the ultimate setup for a Thanksgiving Day disaster. The physics of water and its solid, liquid, and gas phases compels us not to do it.
Caption:“At this time in Mars’ history, we think CO2 is everywhere, in every nook and cranny, and water percolating through the rocks is full of CO2 too,” Joshua Murray says.
Are breakthroughs really a matter of chance, or are they simply waiting to be uncovered by the right person at the right time?
It’s deceptively tricky to distinguish living systems from non-living systems. Physics may be key to solving the problem.
“What modern science has taught us is that life is not a property of matter.”
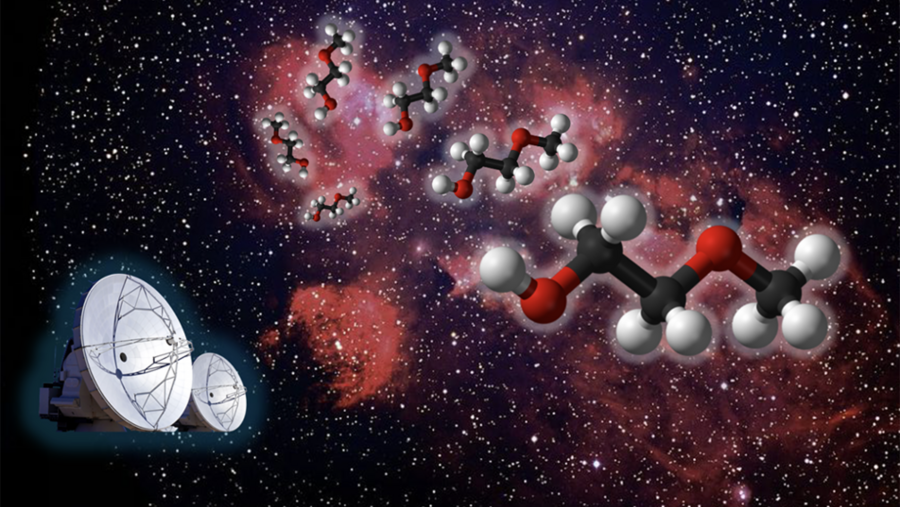
Such discoveries help researchers better understand the development of molecular complexity in space during star formation.
“I hope we take a mindset where we are willing to look for weird life in weird places.”
Claims circulating on the Internet — some from dentists’ websites — suggest toothpaste isn’t necessary for dental health. Is that true?
Plagues, war, and genocide were literally frozen in time.
Although early Earth was a molten hellscape, once it cooled, life arose almost immediately. That original chain of life remains unbroken.
Do the benefits of plastics outweigh the costs?
Our cosmic home, planet Earth, has been through a lot over the past 4.5 billion years. Here are some of its most spectacular changes
Earth wasn’t created until more than 9 billion years after the Big Bang. In some lucky places, life could have arisen almost right away.
While ice itself is slick, slippery, and difficult to navigate across under most circumstances, skaters easily glide across the ice.
Chemists could replace bubbling flasks with tumbling ball mills.
Scientists will be able to make detailed “Claymation-like” movies of chemical reactions.
Capsaicin is already used to treat nerve pain. Early research hints it could do more.
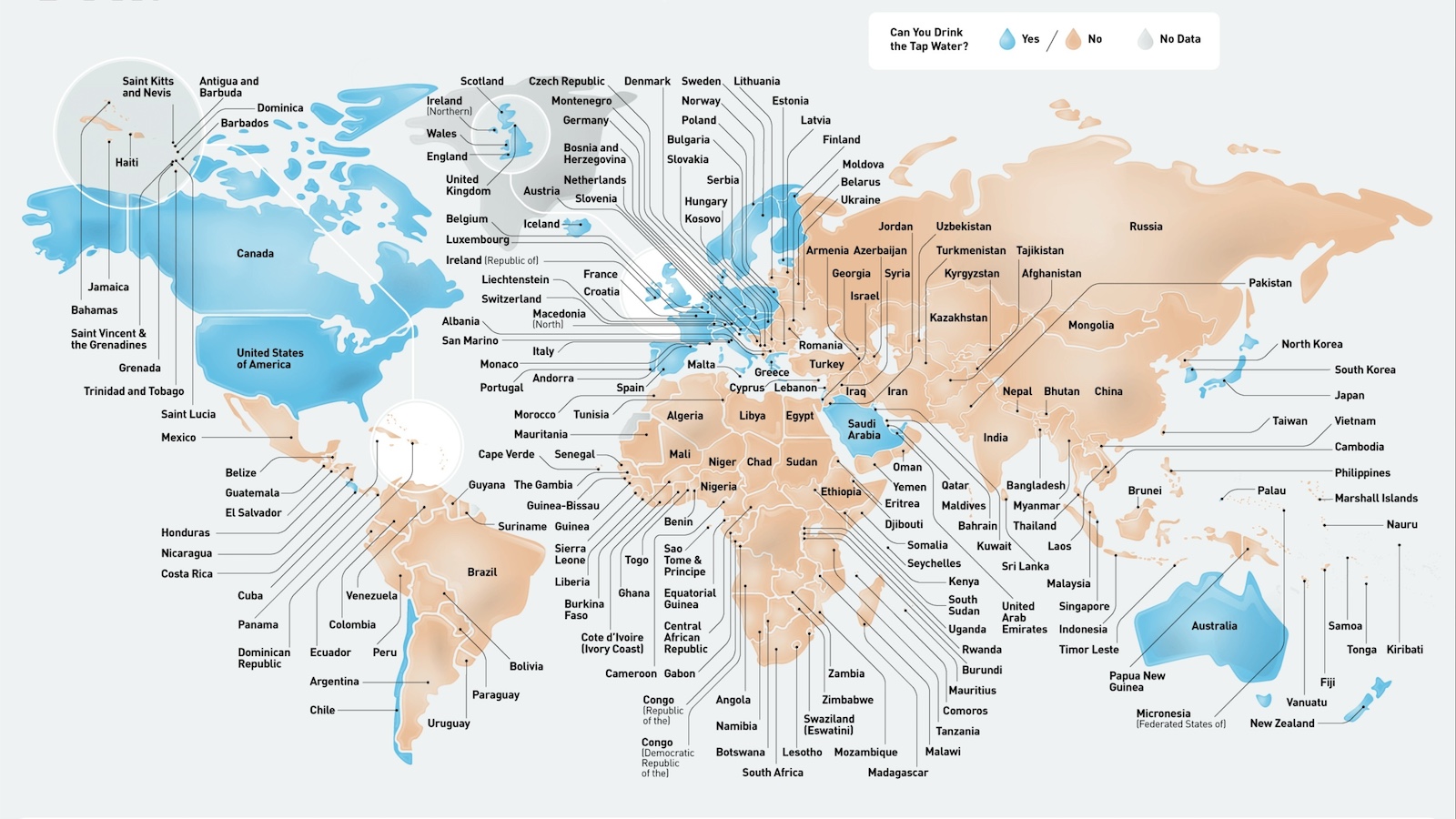
According to the CDC, 50 countries worldwide have drinkable tap water. But look closer, and the picture is more nuanced.
We don’t need to think about what life is made of but rather what it does.
From ancient Greek cosmology to today’s mysteries of dark matter and dark energy, explore the relentless quest to understand the Universe’s invisible forces.
The flavor is “simultaneously fascinating and… abusive.”
Every astrobiologist wants to find an alien. But the public should be skeptical when the “aliens” look like tiny humans.
The best answer we have is, “Life is matter with intentionality.”
Chemical changes inside Mars’ core caused it to lose its magnetic field. This, in turn, caused it to lose its oceans. But how?
Scientists may have detected the somewhat smelly chemical dimethyl sulfide on a planet 120 light-years from Earth.