mice
Scientists watch as mice mouse around an onscreen maze.
It’s more than just weight gain—it’s chronic inflammation and weak immunity.
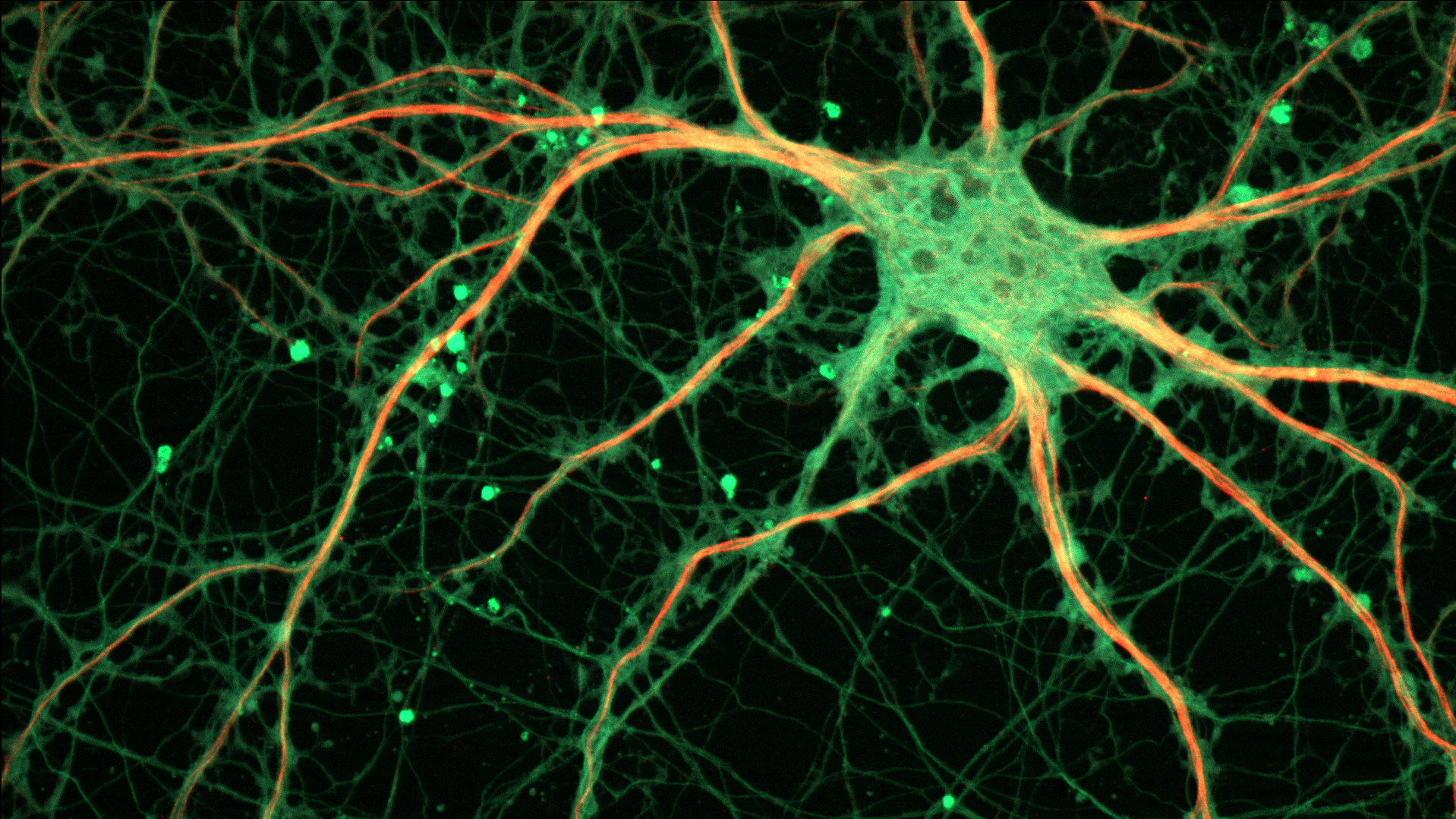
The discovery of a neuron in the brain that acts as the “master controller” of habits could someday change the way we treat addiction and compulsive behaviors.
Scientists are concerned that the results of studies using mice may be affected by gut bacteria.