Throwback Thursday: A Static Electricity Surprise
Add static electricity to the long list of things we thought we understood, but didn’t.
“Electricity can be dangerous. My nephew tried to stick a penny into a plug. Whoever said a penny doesn’t go far didn’t see him shoot across that floor. I told him he was grounded.” -Tim Allen
I know what you’re thinking. “Of course I know what static electricity is!” Surely, you take two initially neutral objects and charge them up — one positively and one negatively — by rubbing them against one another.
Right?
Not so fast! Let’s start off with the electricity you think you know, but prepare yourself: you’re going to be surprised.

You’ve all (hopefully) gotten to play with a Van de Graaff Generator at some point. It’s one of the simplest electricity demonstrations there is. You stand on something insulating (like a milk crate, so you’re not grounded), touch your hands to the generator, have someone turn it on, and your hair (for those of you with hair) stands up on its end!
The reason for this, of course, is that when you turn the Van de Graaff Generator on, the top of it charges up (with positive charges). If you’re connected to it, then (as a pretty good conductor) you charge up with those positive charges as well.
Since positive charges repel one another, those of you with straight enough, long enough hair will notice that the electric forces in your hair easily become more important than gravity or any other electric forces. This causes the very fun phenomenon of causing your hair to stand on end, since positive charges repel other positive charges. (For those of you with very curly hair, the internal electrostatic forces may be stronger than any external charge you can apply; sorry!)

That’s just the simplest case of plain electrostatics, where you give an object (or set of objects) just one type of charge.
But what you’re used to calling static electricity is a little different. You probably think of rubbing two objects together, like your socks on the carpet, or a piece of glass with some silk.

And, as you were (properly) taught, one of these materials loses electrons, leaving it positively charged, while the other one gains electrons, leaving it negatively charged.
This applies to a whole bunch of things, like rubbing a balloon against your hair.

After a good static charging, you’ll notice that the balloon can do all sorts of interesting things: cause your hair to stand up, stick to the wall, or annoy the ever-living-daylights out of your poor grandpa.

How does this happen?
Presumably, you’ve given some extra electrons from the balloon, leaving it negatively charged. And when you bring it close to a neutral object — like a wall — you attract the “opposite” charges on the wall (the nuclei) and repel the “like” charges (the electrons). As long as this configuration remains, the balloon will remain stuck to the wall, as the electric forces, due to static electricity, will hold it in place.
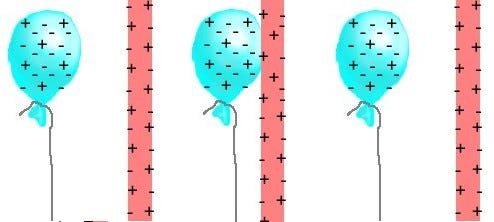
And that’s how you were taught static electricity works.
Simple, right? Unfortunately, it’s too simple! As it turns out, that picture is not quite right. Why not? Imagine what should happen if you take two identical materials, like two sheets of office paper.

If you rubbed two sheets together, you’d expect that neither one would wind up with a static charge on them, right? They’re made of the same material, so neither one should give up negative charges to the other, and so there shouldn’t be any charge. At least, that’s what you’d expect if static electricity worked like we just described it.
Only, that’s not what happens. Let’s take a closer look at this sheet of office paper.
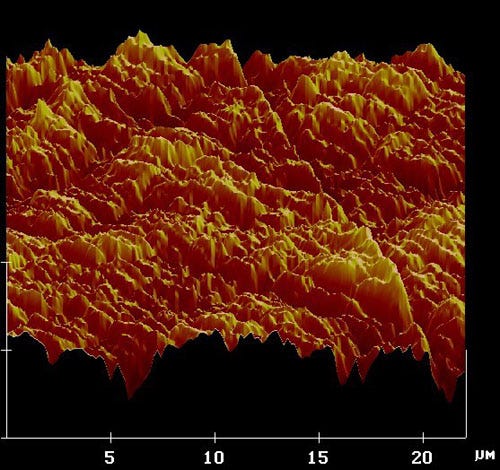
Smooth as paper might seem, at a microscopic level there are tiny imperfections on the surface, visible in the image above at micron-level scales. When you take two of these sheets of paper (or any two identical materials) and rub them together, what do you suppose happens to the voltages on the surface?
Give up?
If you want to know, in science, you need to do the experiment and find out. And amazingly enough, no one had done this experiment until 2011! But thanks to Professor Grzybowski’s group at Northwestern University, we now have the results from this, and they are spectacular.

Instead of not charging, both sheets of paper totally pick up static charge! In fact, different sections of each surface pick up large amount of positive or negative charge. What we’ve been looking at, this whole time, as static electricity, only represents the net charge on these objects, which could be positive, negative or zero. But what actually causes the individual molecules to attract or repel a nearby object has very little to do with the overall charge and everything to do with how those particular nearby molecules are charged! To put it in the authors’ words:
For centuries, it has been assumed that such contact charging derives from the spatially homogeneous material properties (along the material’s surface) and that within a given pair of materials, one charges uniformly positively and the other negatively. We demonstrate that this picture of contact charging is incorrect. While each contact-electrified piece develops a net charge of either positive or negative polarity, each surface supports a random “mosaic” of oppositely charged regions of nanoscopic dimensions. These mosaics of surface charge have the same topological characteristics for different types of electrified dielectrics and accommodate significantly more charge per unit area than previously thought.
So yes, some materials gain electrons and other materials lose electrons when you rub them together. But it is now thought — and this is brand new — that every statically charged material has significant regions of both positive and negative charge!
Not only is this a new finding, it’s now thought that this is the dominant reason for static electricity.

So static electricity, practically, will still work exactly as you expect it to. Only next time you encounter it, you’ll know how it really works!
Leave your comments at our forum, and support Starts With A Bang on Patreon!





