“Impossibly big” bacteria rattle the field of microbiology
- Due to physical limitations, bacterial cells are very small.
- However, a newly discovered species of bacteria is so large that it can be seen with the naked eye. It also contains a DNA-containing nucleus, a structure thought not to exist in bacteria.
- According to bedrock biological theories, these bacteria simply shouldn’t exist.
In 1977, the Monera Kingdom crumbled. It was a young kingdom, only about 50 years old. Consisting of bacteria and similar microbes, it also was the most populated kingdom ever to exist. Scholars often attribute the downfall to Carl Woese, an American microbiologist and biophysicist, who, through genetic analysis, discovered that the borders of the Monera Kingdom were drawn incorrectly.
In addition to crumbling the kingdom, Woese’s finding transformed the Tree of Life, a “family tree” of all species on Earth. Consequently, the former kingdom and its citizenry were separated into two domains — Bacteria and Archaea. And now, a new finding that has rattled the field of microbiology might hint that we are on the verge of another crumbling empire. This time, it is due to a species of unusually big bacteria named the “Magnificent Sulfur Margarita.”
Thiomargarita magnifica: the bacterium that should not exist
Technically, this species of big bacteria is named Thiomargarita magnifica (which could also be translated “Big Sulfur Pearl,” but Margarita is cooler). A couple years ago, however, it did not have a name. Only a handful of scientists knew it existed, and they had no idea it was a bacterium.
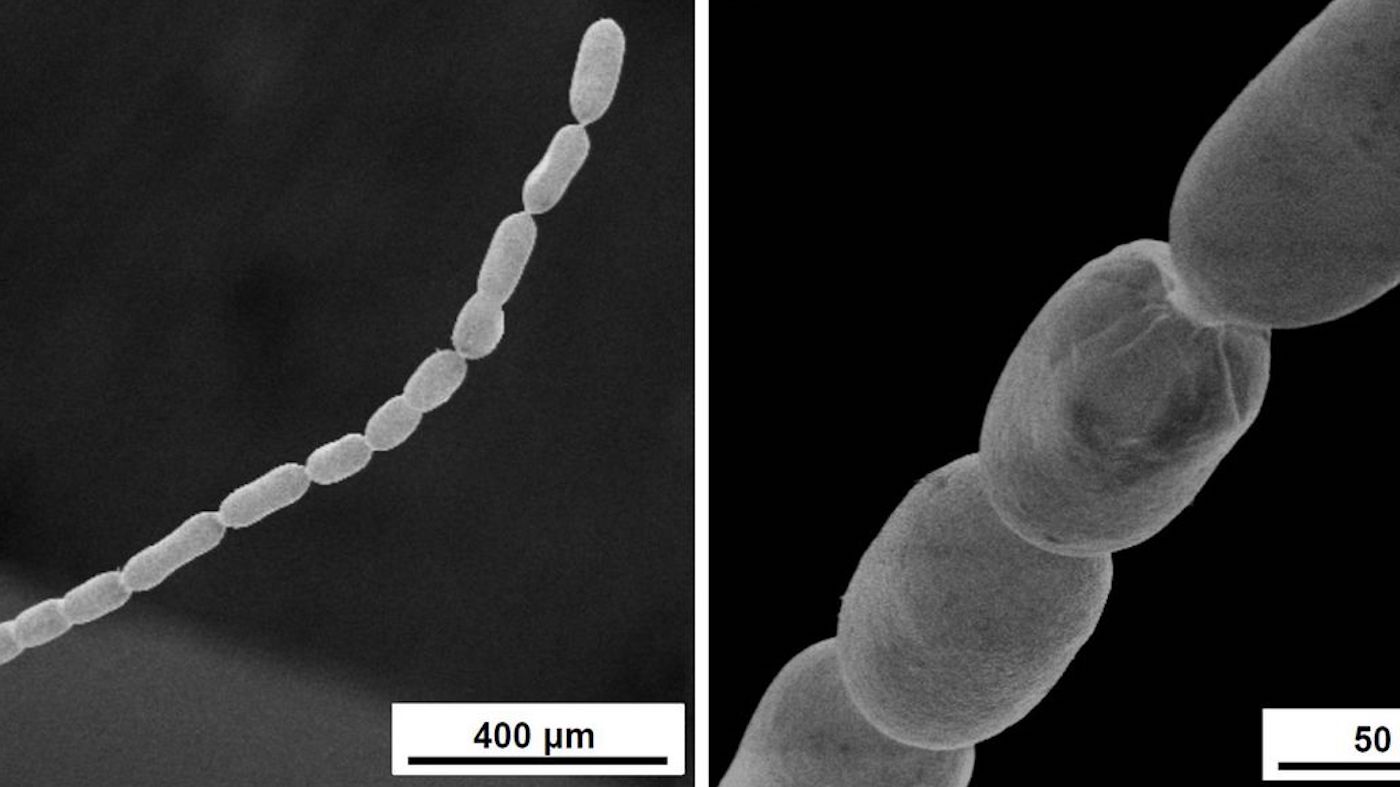
Olivier Gros, a marine biologist at the University of the French Antilles, noticed centimeter-long white strands attached to submerged mangrove leaves. At the time, he suspected it was fungi but did not recognize the species. Only about seven percent of fungi have been characterized, so Gros collected a sample and took it back to his lab. Gros’ graduate student, Jean-Marie Volland, began analyzing the unusual organism. And things got weird.
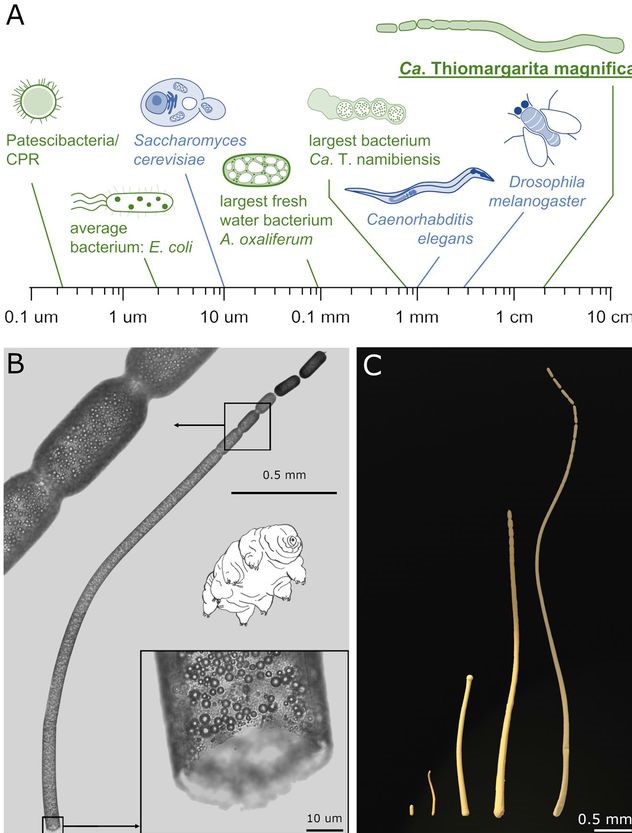
According to the study, which is still in preprint and needs to be verified by peer review, early experiments suggested the organism was a denizen of the Bacteria Domain, but it was 50 times larger than the biggest bacterium ever discovered — big enough to see without a microscope. That is supposed to be impossible — or so it would seem based on the diffusion-constraint hypothesis, which posits that bacteria are size-limited due to the rate of nutrient diffusion.
Bacterial biophysics
Put into English, the diffusion-constraint hypothesis essentially states, “If a cell wants to move something over a long distance, it has to do some work.” How much work needs to be done? It depends on the size of the cell.
Imagine putting a drop of black dye at the edge of a shot glass full of water. Within a few seconds, the dye has spread to the center of the glass. This is passive diffusion. It works great for small cells, like bacteria. Now, imagine putting a drop of black dye at the edge of an Olympic-size swimming pool. The dye will take way too long to get to the center, and it will be far too diluted when it does — unless a swimmer scoops up the dye with a cup and swims it to the center of the pool. This is active diffusion. It takes work, which requires energy.
But bacteria are lazy, so they rely only on passive diffusion. They absorb nutrients through their membranes and then let the laws of physics take over. Just like the swimming pool, if the bacterium becomes too big, the nutrients cannot spread efficiently throughout the cell. Larger and more complex cells known as eukaryotic cells, such as human cells, overcome this physical limitation by using elaborate transport systems that shuttle nutrients to where they are needed. These systems don’t exist in bacteria.
How the big bacteria break the rules
The massive microbe has found a way to get around the diffusion-constraint hypothesis: It contains a fluid-filled sac, which squishes the intracellular contents up against its outer cell wall. (Returning to the previous analogy, this would be like putting a gigantic balloon in the swimming pool, forcing all the water to the edges.) This means that nutrients do not have to diffuse very far, as they are limited to the edges of the bacterium.

The big bacteria break another big rule: they have something resembling a nucleus, a special DNA-containing compartment that is the defining structure of eukaryotic cells. A nucleus is what, according to endosymbiotic theory, separates simpler prokaryotic cells (Domains Bacteria and Archaea) from the more complex eukaryotic cells. Somehow, these big bacteria have taken a very strange evolutionary turn, one that would not be predicted by any existing theory. In other words, these big bacteria shouldn’t exist.
Is the Tree of Life wrong?
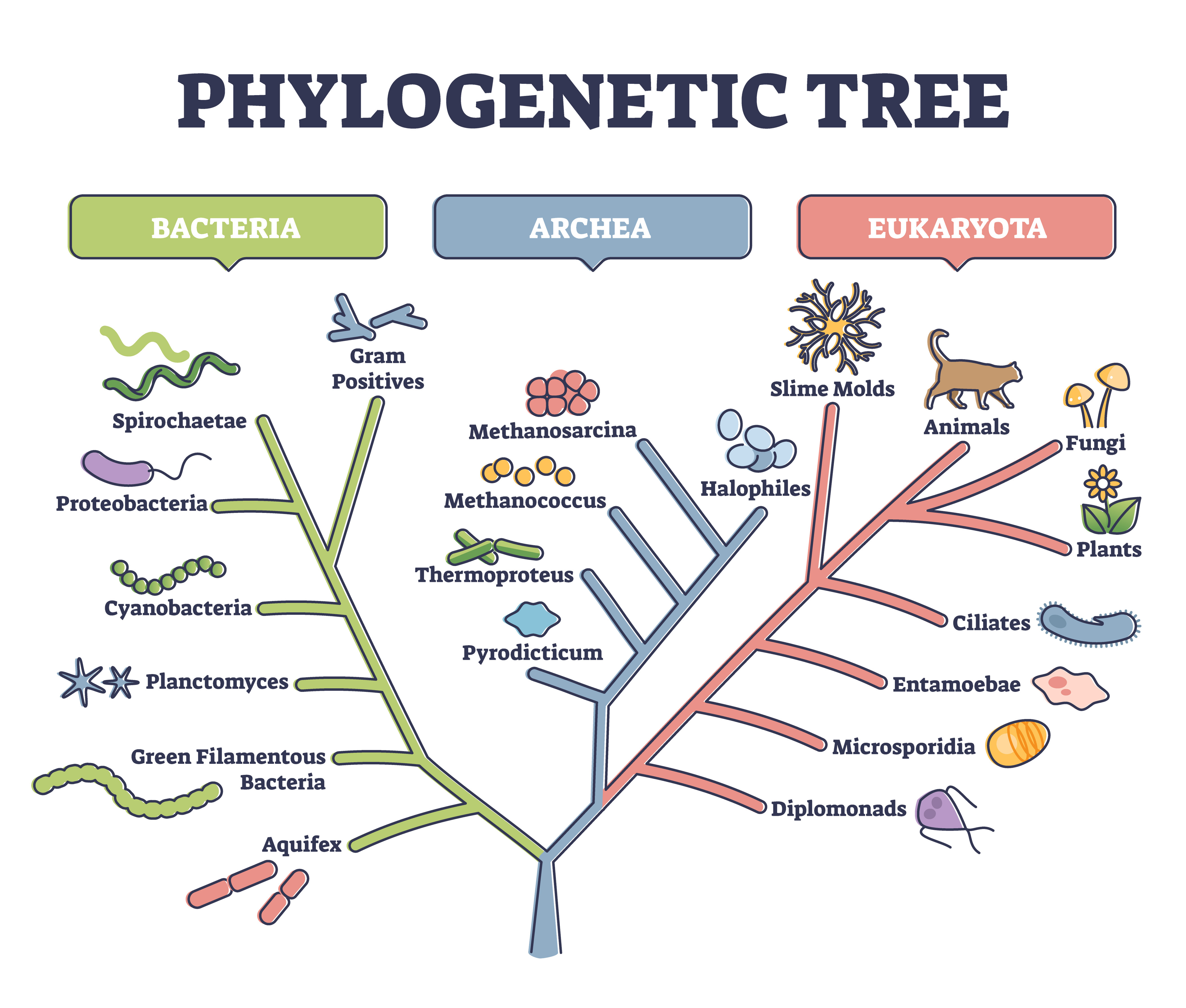
The so-called “Tree of Life,” which depicts our understanding of evolutionary relationships going back to the very beginning — that is, the origin of life — is a bedrock principle taught in biology textbooks across the world. Is it wrong?
Probably not, but the big bacteria strongly suggest that our understanding of the evolution of biological complexity is woefully incomplete. They take their place alongside giant viruses, some of which are bigger than bacteria. Together, these anomalous microbes hint that a mystery remains deep within the heart of evolutionary biology.