Cancer evolution is mathematical
Cancer is often seen as a disease that arises from genetic mutations causing cells to divide uncontrollably and invade other parts of the body. But the spread of cells away from their origins is actually a normal process in some cases. The embryo burrows into the uterus during early pregnancy. Immune cells spread from lymph nodes to sites of infection to attack the invading bacteria. And germ cells migrate to where the gonad will be during early human development.
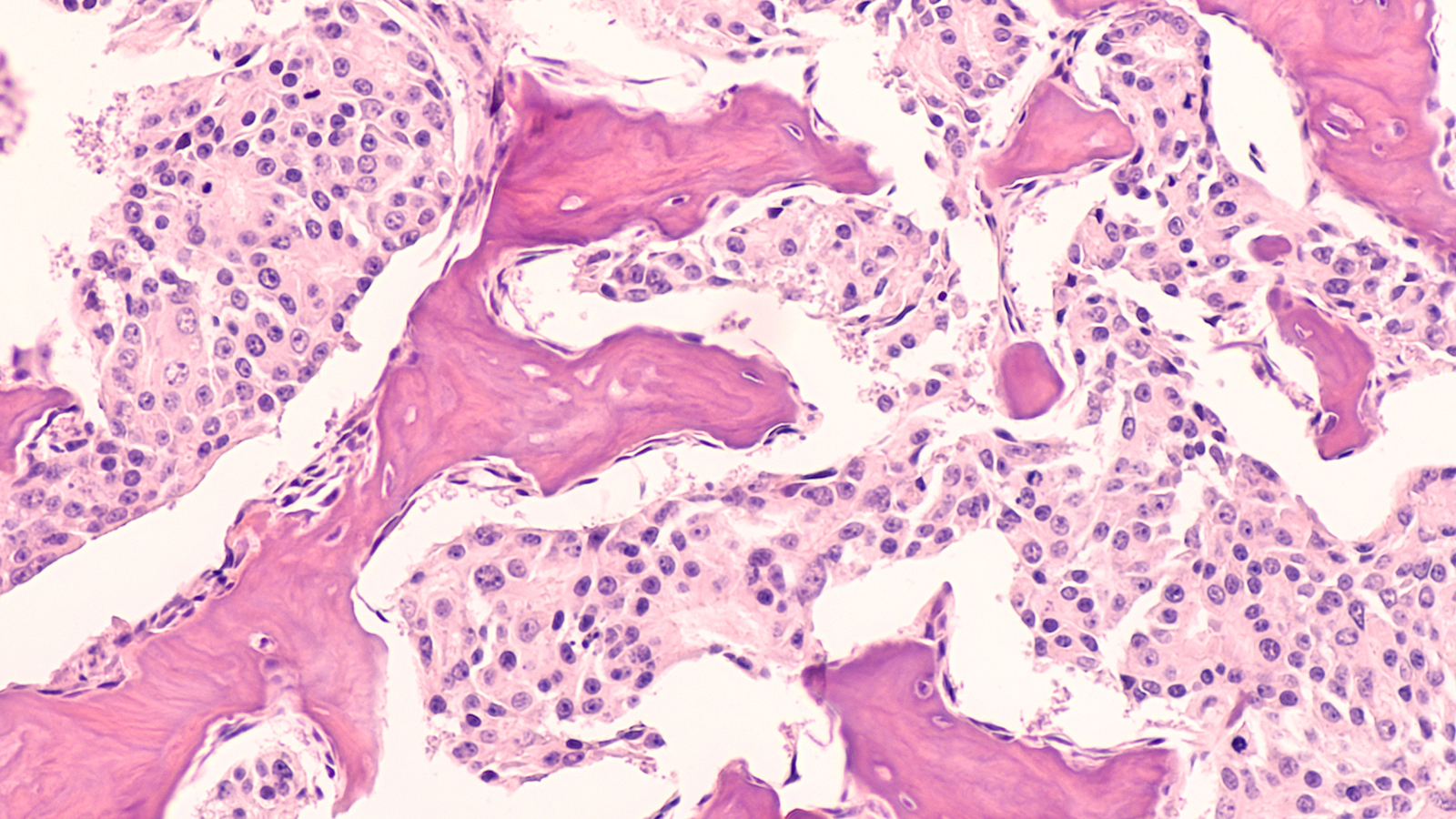
Cancer is not a uniform disease. Rather, cancer is a disease of phenotypic plasticity, meaning tumor cells can change from one form or function to another. This includes reverting to less mature states and losing their normal function, which can result in treatment resistance, or changing their cell type altogether, which facilitates metastasis.
In addition to direct changes in your DNA in cancer, a key driver of cancer progression is where and when your DNA is activated. If your DNA contains the “words” that spell out individual genes, then epigenetics is the “grammar” of your genome, telling those genes whether they should be turned on or off in a given tissue. Even though all tissues in the body have almost exactly the same DNA sequence, they can all carry out different functions because of chemical and structural modifications that change which genes are activated and how. This “epigenome” can be influenced by environmental exposures such as diet, adding a dimension to how researchers understand drivers of health beyond the DNA code inherited from your parents.
I’m a cancer researcher, and my laboratory at Johns Hopkins University studies how the differences among normal tissues are controlled by an epigenetic code, and how this code is disrupted in cancer. In our recently published review, colleague Andre Levchenko at Yale University and I describe a new approach to understanding cancer plasticity by combining epigenetics with mathematics. Specifically, we propose how the concept of stochasticity can shed light on why cancers metastasize and become resistant to treatments.
What is stochasticity?
Stochasticity is a mathematical concept that refers to the idea that the randomness of the steps in a process affects the predictability of its outcome. Albert Einstein famously studied this concept applied to the movement of particles suspended in a liquid or gas. Researchers can apply stochasticity to study the spread, resistance and evolution of COVID-19, the behavior of the stock market and almost any game inside a casino.
A key way to measure the stochasticity of a process is entropy, which quantifies the degree of uncertainty in an outcome. For example, a fair coin toss has an entropy of one, or low information, because there is no way to predict whether the coin toss will be heads or tails. But a weighted coin toss has an entropy of zero, or high information, as the outcome is already known and no new information will be gained by tossing the coin.
Researchers can use entropy to measure the amount of informational noise in telecommunications. Entropy can also help players beat the game Wordle. The word with highest entropy and thus the largest expected new information after each guess would be your best bet.
Epigenetics ties stochasticity and cancer together
Incorporating stochasticity and entropy into biology allows researchers to better understand phenotypic plasticity in cancer. This could even reconceptualize development by including reversibility, or moving against “time’s arrow.” This departs from a more classic perspective of embryonic development that views tissues as becoming progressively and irreversibly differentiated into their final state as they develop.
Experimental and computational biologists are using entropy to understand the underlying randomness in how cells are internally organized, respond to environmental signals and mature and form tissues.
Stochasticity in epigenetics is critical to how cancer evolves. For example, a condition called Barrett’s esophagus occurs when healthy cells in the esophagus develop features more like what cells lining the intestine normally have, which can ultimately lead to esophageal cancer. This is caused by progressive random changes in the epigenetic code, and this shift occurs more rapidly once it reaches a certain threshold. The stochastic nature of these epigenetic changes also leads to increased entropy in the function of those genes, and progression toward cancer.
By measuring the gene activity and epigenetic changes of individual cells, biologists and mathematicians can compare the entropy in cancer cells with the normal cells surrounding them. Scientists are now beginning to identify regions of the genome that mediate stochasticity in cancer. A study that has not yet been peer-reviewed found that entropy is related to how chromosomes are physically compacted in the nucleus, another key epigenetic mechanism for controlling gene activity in cancer.
There is a connection between entropy and aging as well. My colleagues and I found that human aging is associated with an increase in epigenetic entropy in sun-damaged skin. Parts of the genome that have high entropy experience further loss of epigenetic information in sun-exposed skin, which can lead to cancer. Recently, researchers have identified DNA damage as a cause for this age-associated entropy in mice. Thus, if epigenetic entropy increases in aging and is related to DNA damage, it might help explain why cancer risk starkly increases with age.
By identifying how epigenetic entropy triggers cancer, scientists might be able to better detect cancer at its early stages, and design drugs that reduce entropy and so decrease the risk of tumors spreading and becoming resistant to treatment.
And perhaps most importantly, epigenetic entropy shows that you can’t fully understand cancer without mathematics. Biology is catching up with other hard sciences in incorporating mathematical methods with biological experimentation.
This article is republished from The Conversation under a Creative Commons license. Read the original article.