Venus Is Dead! New Analysis Shows Phosphine, A Possible Biosignature, Is Absent

If there’s no phosphine, there’s no indirect evidence for biological activity there.
In one of the biggest surprises in the history of planetary science, a September 2020 study announced the presence of phosphine gas in Venus’s cloud decks: a tantalizing hint that could be due to biological processes. Phosphine (PH3), which is chemically similar to ammonia (NH3) except with phosphorus standing in for nitrogen, is only produced naturally on Earth by biochemical processes, but was claimed to exist on Venus at the 20 parts-per-billion level. With two separate observatories claiming this detection — both the JCMT and ALMA — the signal appeared to be quite robust.
The discovery raised the exciting possibility that Venus’s clouds are home to life, and that those biological life forms are producing this phosphine. Indeed, photochemical processes from the Sun’s radiation, chemical processes from thermodynamic atmospheric effects, and geochemical processes from surface chemistry reactions were all deemed insufficient for producing these levels of phosphine.
So, is it life? Maybe. But that’s assuming phosphine is actually present, and a new study has just been submitted that calls the entire detection into doubt. Here’s what it means, and why the evidence that Venus might be anything other than a completely dead, hellish planet might have just evaporated entirely.

Back in September, a team of scientists led by Professor Jane Greaves announced the discovery of phosphine by applying a well-known and very straightforward technique in astronomy.
- They measured light in the radio part of the spectrum,
- modeled the known components and conditions of the atmosphere from prior observations,
- made a prediction for what signals should appear when the light was broken up into individual wavelengths,
- made a prediction for what features should be present (or absent) if phosphine were actually there (or not),
- and then break that light up, spectroscopically, to analyze that light to determine the presence and abundance of phosphine.
What they saw, in two independent data sets from both JCMT and ALMA, was an absorption feature at the same wavelength and of the same signal strength, indicating that something was there causing that radio light to be absorbed at a particular frequency. That frequency corresponds to a natural transition in phosphine molecules. If this analysis was performed correctly, it means that phosphine is really there.
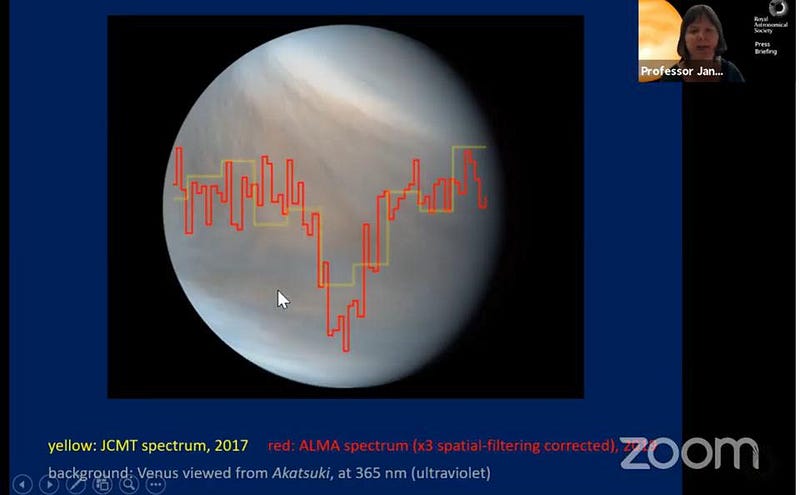
Immediately and quite understandably, this launched a firestorm of speculation. How could this phosphine have been formed? What could be causing its presence, which appeared strongest at mid-latitudes and at altitudes of between 40 and 60 km: in or near the “temperate zone” on Venus, where temperatures and atmospheric pressures are similar to those on Earth’s surface?
Well, phosphine naturally exists on Earth because bacteria in anaerobic environments — speculated to be a form of E. coli — produce it. Venus’s atmosphere is an anaerobic environment, and the mechanisms that produce it in gas giants like Jupiter and Saturn wouldn’t work under Venusian conditions. We can produce it artificially on Earth via controlled chemical reactions, but those conditions don’t apply on Venus, either.
Perhaps, the discovery team speculated, it was indeed because of organisms in Venus’s cloud decks at specific altitudes, which could create enough phosphine if their density was close to the limit of what’s maximally possible.
But there’s another possible explanation that — if we’re being responsible scientists — we have to consider. Perhaps there’s a flaw in how the data was analyzed, and perhaps that means that phosphine isn’t present after all. Before we run down the route of speculation as to what might be causing this molecule’s existence, from biochemical to thermodynamic to inorganic chemistry pathways, we have to make certain that we aren’t fooling ourselves entirely. Even when we’re being as responsible as possible, we have to rule out the possibility that our analysis has led us to a mistaken conclusion.
The first reasonable check we can make is to look at older data: from missions that have probed Venus’s atmosphere in the past. In 1978, the Pioneer-Venus Large Probe Neutral Mass Spectrometer was launched, taking measurements of masses of neutral gases at different altitudes within Venus’s clouds. In particular, they were sensitive to gases between 50–60 km, which is right in the zone where the recent data supports the existence of phosphine. Less than 2 weeks after the phosphine-on-Venus announcement was made, a team re-examined that data to see if it was consistent with phosphine or not.

What they found, by breaking up the components of Venus’s atmosphere by mass, was that:
- atomic phosphorus (P) is present, unambiguously,
- a signal indicating either phosphine (PH3) or a hybrid of it with hydrogen sulfide (H2S) is present,
- and deuterated phosphine (PH2D), where one of the hydrogen atoms in phosphine has a neutron in its nucleus, is also present unambiguously.
Atomic phosphorus could be a fragment of phosphine, and other fragments (such as PH and PH2) may have been detected as well. Previously, the signature that could be phosphine was attributed to hydrogen sulfide, but this reanalysis suggests that phosphine might compose part or even all of that signal. Would the amounts be consistent with what the Greaves et al. study suggests, though? According to the authors:
“While intensities of the peaks are low, they are perhaps consistent with the ~20 ppb abundances reported by Greaves et al.”
So, perhaps. The older data doesn’t give us a slam dunk, but it doesn’t disfavor phosphine, either.
What about other observations? We’ve been monitoring Venus in the infrared part of the spectrum for many years, which reveals a different set of molecular signatures at different altitudes than radio observations reveal: probing the cloud-tops of the Venusian atmosphere. Since 2012, those observations have allowed us to determine the presence and distribution of both sulfur dioxide (SO2) and water molecules (H2O).
If phosphine is present on Venus, and was generated by the mechanism that the authors proposed, it should occasionally rise up to the cloud-tops, making an appearance in the infrared spectrum. By examining the data that exists, spanning years, a team of scientists — which happens to include Greaves herself on the author list — they found only one window, in March of 2015, where a spectral signature consistent with phosphine arises. The authors conclude:
“From the absence of any feature at this frequency, we derive [a 99.7%-confident] upper limit of 5 ppb…”
meaning that they disfavor the conclusions drawn by the radio data, instead finding that,
“Our upper limit is not compatible with a constant mixing ratio of 20 ppb throughout the mesosphere, as announced by Greaves et al. (2020).”

But the most direct refutation would be if it turned out that the high-quality ALMA data — which provided the strongest evidence for phosphine by far — didn’t indicate the presence of phosphine, after all. This would be a remarkable refutation, as the original study claimed to detect a spectral line that was consistent with phosphine, unambiguously, at an absurdly high ~15-sigma confidence level. The odds of a ~15-sigma result being wrong are about as good as having a lottery the size of the observable Universe and picking, with one choice, the winning subatomic particle.
On October 19, 2020, a paper was submitted to the arXiv: the preprint server where almost all astrophysics papers go before they’re published. By doing an independent reanalysis of the raw ALMA data themselves, they found no trace of such a signal with any comparable confidence. In fact, their big findings can be summarized in two points:
- there was a flaw in how the original team dealt with the data, which they demonstrate leads to erroneously high-confidence signals,
- and that when they (correctly) deal with the data, the most “confident” signal that arises is not even at ~2-sigma confidence, which qualifies as a non-detection.
Let’s look at each point, as explained by the paper’s authors, in a little more depth.
The flaw in the analysis. What you typically do when you perform spectroscopy on a planet, as an astronomer, is the following:
- your instrument takes data,
- you process the data according to a processing script that’s specific to that instrument,
- you throw out the data where the instrument is noisy (too close to the limb of the planet, for example),
- you “bin” the data by collecting it into a series of narrow, consecutive intervals,
- you subtract out the continuum (the background, base level of signal or noise across all bins),
- you model the instrumental ripples around (but not including) the region where you expect the signal to appear,
- you subtract the best-fit of the modeled region from the region-of-interest (more on this in a second),
- and then you analyze what’s left to look for the presence or absence of particular signatures.
The problem is that the next-to-last step is powerful, but potentially dangerous. In general, you hope to remove noise structures and instrumental effects, while leaving the real spectral features intact. That’s what the original (Greaves et al.) team did, and that’s how they arrived at their detection of phosphine.
But what the reanalysis team found, led by Ignas Snellen, was that the fit-and-subtraction method used by the Greaves et al. team leads “to the severe overestimations of the significance of spectral features and artificial results.” In other words, the claimed detection of phosphine is unreliable, and they go on to demonstrate how.
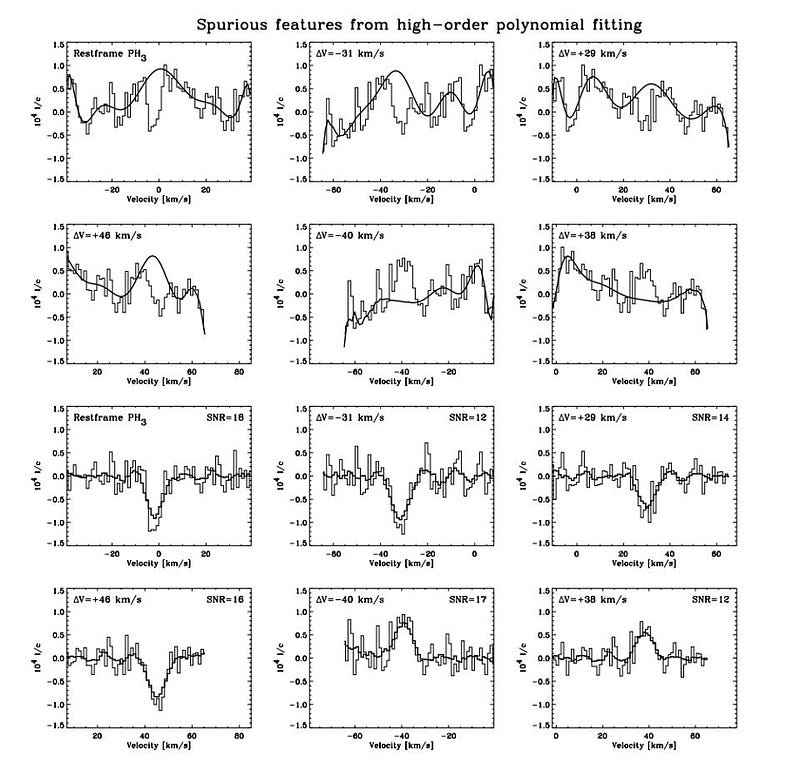
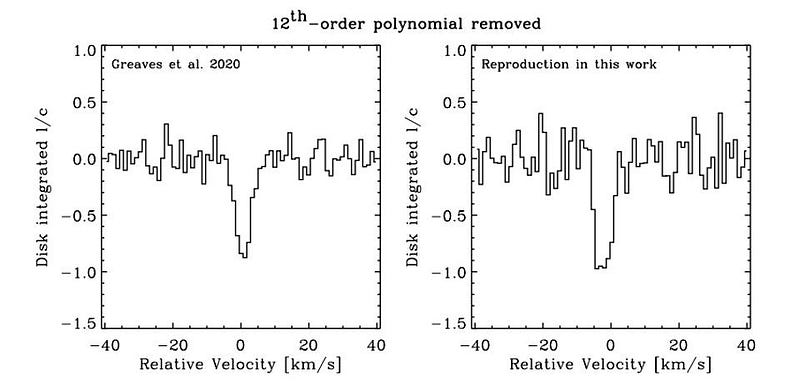
The detection signal “disappears” without improper analysis techniques. The above graphic is “figure 2” from the Snellen et al. paper, which you should think of in two halves. The first half shows six regions of the spectrum: after continuum subtraction but before modeling the instrument ripples. The raw data, shown in narrow bar graph style, has the “best-fit” model overlaid atop it: the smooth, continuous curve.
The second half shows those same six regions, but with that “best-fit” model subtracted from it. As you can clearly see, absorption features stick out in all six regions like a sore thumb.
But here’s the problem: the upper-left of those six boxes corresponds to the region where phosphine was claimed to be detected; the other five boxes correspond to absolutely nothing. All six regions have enormously high signal-to-noise ratios. And the conclusion should be the same for all six regions: there’s nothing there. According to Snellen’s paper:
“It shows that the procedure followed by [Greaves et al.] is incorrect, and results in spurious, high signal-to-noise lines.”

Instead, Snellen’s team argues, it’s more responsible to treat the data with a much gentler noise-removal procedure: one that won’t result in spurious, high signal-to-noise lines. When that’s done, the signal-to-noise ratio is only a factor of ~2; without any noise removal at all, the signal-to-noise ratio is only ~1. As Snellen’s team correctly points out, “In astronomy, features at such a low [signal-to-noise ratio] are generally deemed not statistically significant.”
So, what does this all mean? It means that the initial detection of phosphine, as claimed by the Greaves et al. team, is suddenly thrown into tremendous doubt. If they’ve analyzed their data in an unsound fashion, as this new work contends, then this chemical signature might not be present, after all. This new paper still needs to go through the peer review process, and I suspect that the original team will perform a reanalysis themselves to see what conclusions they arrive at.
In the absence of what initially appeared to be extraordinary evidence, we need to return to our default position: Venus has no evidence for being a biologically active world. Unless this new analysis is refuted and the initial work is vindicated, the idea of phosphine on Venus may be as dead as Venus itself.
Starts With A Bang is written by Ethan Siegel, Ph.D., author of Beyond The Galaxy, and Treknology: The Science of Star Trek from Tricorders to Warp Drive.





